What is diabetic ketoacidosis?

diabetic ketoacidosis
DIABETIC KETOACIDOSIS ( DKA)
DKA is a potentially life-threatening complication of diabetes that occurs when the body begins to break down fats for energy instead of sugars.
In people without diabetes, insulin helps sugar enter the cells, which use it for fuel. A person with diabetes does not make enough insulin to properly transport the sugar, which means that their body cannot use it for energy.
Without enough sugar, the liver turns some of the fat in the body into acids called ketones. Ketones build up in the bloodstream and spill over into the urine. When these excess ketones get into the blood, the blood becomes acidic, causing DKA.
DKA is a medical emergency. Anyone living with diabetes needs to learn the signs and symptoms of DKA to know when to seek urgent medical treatment.
DKA IN TYPE 1 VS TYPE 2 DIABETES
DKA is much more likely to occur in people with type 1 diabetes, but anyone with type 1 or type 2 diabetes can develop it. Those who need to take insulin have a more severe form of diabetes and, therefore, will have a higher risk of DKA.
People with type 2 diabetes who may be prone to ketosis include:
- older people
- people who are overweight
- people from non-white ethnic groups
SYMPTOMS OF DKA

SYMPTOMS OF DKA LIKE WEAKNESS Diabetic ketoacidosis symptoms often start suddenly and can include:
- excessive thirst
- nausea
- vomiting
- frequent urination
- abdominal pain and discomfort
- rapid breathing
- a flushed face
- fatigue
- weakness
- confusion
- fruity-smelling breath
- dry mouth and skin
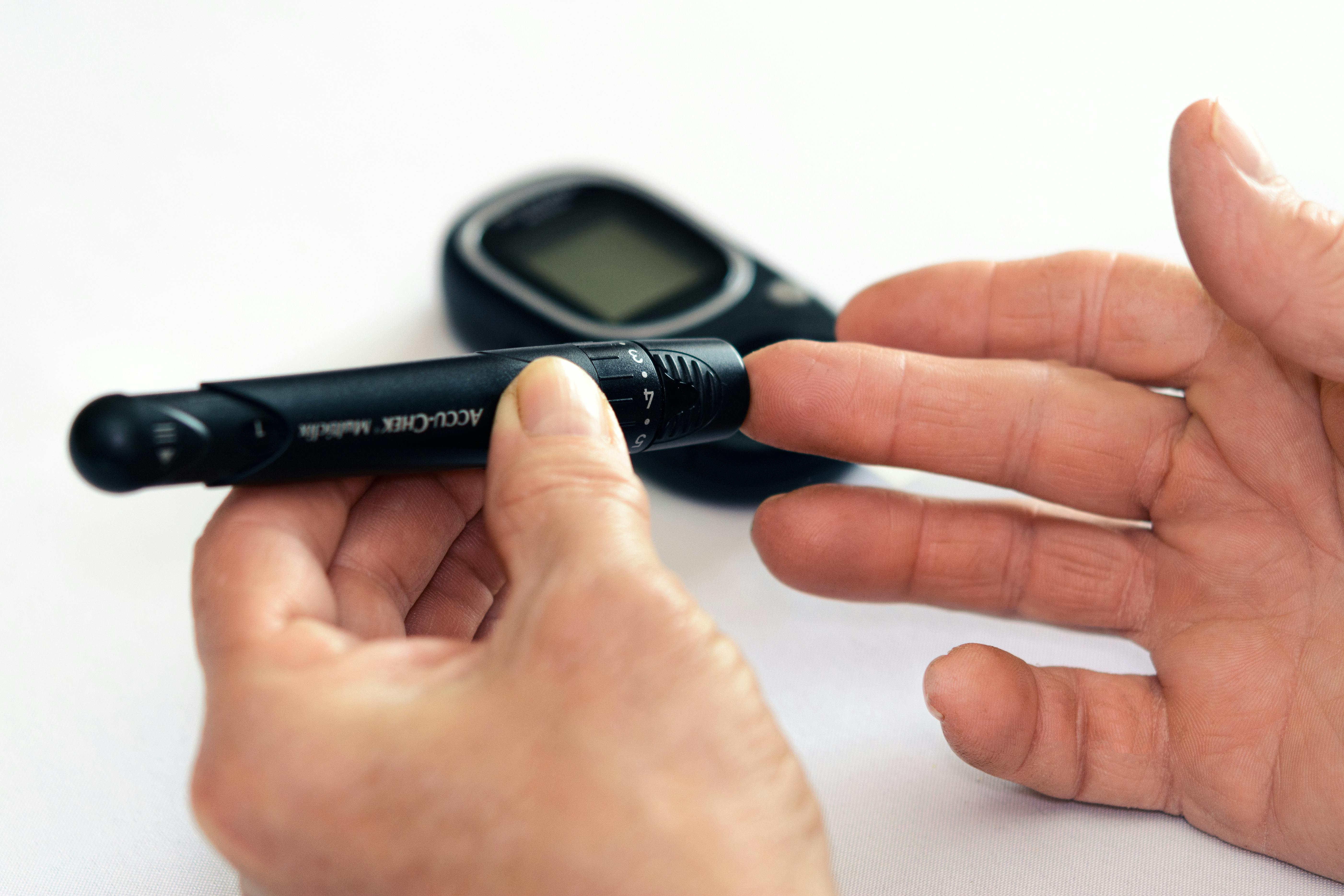
People with diabetes who monitor their blood sugar regularly may also find that their blood sugar readings have become very high.
For others, the symptoms of DKA may be the first indication of diabetes, which may lead to a diagnosis.
CAUSES

- intravenous (IV) fluid replacements
- IV insulin drip until blood sugar levels drop to within an acceptable range
- IV electrolyte replacements very high blood sugar levels and low levels of insulin cause diabetic ketoacidosis. Either illness or a problem with insulin therapy can put a person at risk of having high blood sugar or low insulin, even with regular diabetes treatment. Illness and infection alter the production of some of the body’s hormones, such as cortisol and epinephrine. These hormones change how insulin works in the body and can reduce its effectiveness, which some people may need to counter by taking extra insulin while they are unwell. Problems with prescribed insulin therapy can also cause DKA. Some issues with insulin therapy that may trigger DKA include:
- missing an insulin shot
- a clogged insulin pump
- not using the correct insulin dosage
Other triggers for DKA can include:
- a heart attack
- a stroke
- physical trauma, such as a car accident
- emotional stress
- drug or alcohol misuse
- certain medications
People with type 1 diabetes or those who frequently miss insulin doses have the highest risk of developing DKA. Some diabetes medications may increase the risk of DKA even when the blood sugar is not high.
Treatment

-
When treating diabetic ketoacidosis, doctors aim to normalize blood sugar levels. They may prescribe the following treatments:
- The treatment for DKA can sometimes cause complications. Possible complications include:
- hypoglycemia, or low blood sugar, from insulin, rapidly entering the body
- hypokalemia, or low potassium levels, from fast fluid replacement
- cerebral edema, or swelling in the brain, as a result of changing the blood sugar levels too quickly
Due to the risk of these complications, doctors will closely monitor people with DKA in the hospital.
Prevention
Attacks of DKA can be prevented in those known to have diabetes to an extent by adherence to “sick day rules“; these are clear-cut instructions to people on how to treat themselves when unwell.
Instructions include advice on how much extra insulin to take when sugar levels appear uncontrolled, an easily digestible diet rich in salt and carbohydrates, means to suppress fever and treat infection, and recommendations on when to call for medical help.
People with diabetes can monitor their ketone levels when unwell and seek help if they are elevated.
Management

MAIN AIM OF TREATMENT The main aim in the treatment of diabetic ketoacidosis is to replace the lost fluids and electrolytes while suppressing the high blood sugars and ketone production with insulin. Admission to an intensive care unit (ICU) or similar high-dependency area or ward for close observation may be necessary.
Fluid replacement
The amount of fluid replaced depends on the estimated degree of dehydration. If dehydration is so severe as to cause shock (severely decreased blood pressure with insufficient blood supply to the body’s organs), or a depressed level of consciousness, rapid infusion of saline (1 liter for adults, 10 ml/kg in repeated doses for children) is recommended to restore circulating volume. Slower rehydration based on calculated water and sodium shortage may be possible if the dehydration is moderate, and again saline is the recommended fluid. Very mild ketoacidosis with no associated vomiting and mild dehydration may be treated with oral rehydration and subcutaneous rather than intravenous insulin under observation for signs of deterioration.
Normal saline (0.9% saline) has generally been the fluid of choice. There have been a few small trials looking at balanced fluids with few differences.
A special but unusual consideration is cardiogenic shock, where the blood pressure is decreased not due to dehydration but due to the inability of the heart to pump blood through the blood vessels. This situation requires ICU admission, monitoring of the central venous pressure (which requires the insertion of a central venous catheter in a large upper body vein), and the administration of medication that increases the heart-pumping action and blood pressure.
Insulin
Some guidelines recommend a bolus (initial large dose) of insulin of 0.1 units of insulin per kilogram of body weight. This can be administered immediately after the potassium level is known to be higher than 3.3 mmol/l; if the level is any lower, administering insulin could lead to a dangerously low potassium level (see below).[ Other guidelines recommend delaying the initiation of insulin until fluids have been administered. It is possible to use rapid-acting insulin analog injections under the skin for mild or moderate cases.
In general, insulin is given at 0.1 units/kg per hour to reduce blood sugars and suppress ketone production. Guidelines differ as to which dose to use when blood sugar levels start falling; some recommend reducing the dose of insulin once glucose falls below 16.6 mmol/l (300 mg/dl) but others recommend infusing glucose in addition to saline to allow for ongoing infusion of higher doses of insulin.

Potassium
Potassium levels can fluctuate severely during the treatment of DKA because insulin decreases potassium levels in the blood by redistributing it into cells via increased sodium-potassium pump activity. A large part of the shifted extracellular potassium would have been lost in urine because of osmotic diuresis. Hypokalemia (low blood potassium concentration) often follows treatment. This increases the risk of dangerous irregularities in the heart rate. Therefore, continuous observation of the heart rate is recommended, as well as the repeated measurement of the potassium levels and addition of potassium to the intravenous fluids once levels fall below 5.3 mmol/l. If potassium levels fall below 3.3 mmol/l, insulin administration may need to be interrupted to allow correction of hypokalemia.
Sodium bicarbonate
The administration of sodium bicarbonate solution to rapidly improve the acid levels in the blood is controversial. There is little evidence that it improves outcomes beyond standard therapy, and indeed some evidence that while it may improve the acidity of the blood, it may worsen acidity inside the body’s cells and increase the risk of certain complications. Its use is therefore discouraged, although some guidelines recommend it for extreme acidosis (pH<6.9), and smaller amounts for severe acidosis (pH 6.9–7.0).
Cerebral edema
Cerebral edema, if associated with coma, often necessitates admission to intensive care, artificial ventilation, and close observation. The administration of fluids is slowed. The ideal treatment of cerebral edema in DKA is not established, but intravenous mannitol and hypertonic saline (3%) are used—as in some other forms of cerebral edema—in an attempt to reduce the swelling.
Resolution
The resolution of DKA is defined as a general improvement in the symptoms, such as the ability to tolerate oral nutrition and fluids, normalization of blood acidity (pH>7.3), and absence of ketones in the blood (<1 mmol/l) or urine. Once this has been achieved, insulin may be switched to the usual subcutaneously administered regimen, one hour after which the intravenous administration can be discontinued.
In people with suspected ketosis-prone type 2 diabetes, the determination of antibodies against glutamic acid decarboxylase and islet cells may aid in the decision whether to continue insulin administration long-term (if antibodies are detected), or whether to withdraw insulin and attempt treatment with oral medication as in type 2 diabetes. Generally speaking, routine measurement of C-peptide as a measure of insulin production is not recommended unless there is genuine doubt as to whether someone has type 1 or type 2 diabetes.
Epidemiology
Diabetic ketoacidosis occurs in 4.6–8.0 per 1000 people with diabetes annually. Rates among those with type 1 diabetes are higher with about 4% in the United Kingdom developing DKA a year while in Malaysia the condition affects about 25% a year. In the United States, 135,000 hospital admissions occur annually as a result of DKA, at an estimated cost of $2.4 billion or a quarter to half the total cost of caring for people with type 1 diabetes. There has been a documented increasing trend in hospital admissions.

EATING DISORDER The risk is increased in those with an ongoing risk factor, such as an eating disorder, and those who cannot afford insulin. About 30% of children with type 1 diabetes receive their diagnosis after an episode of DKA. Lower socioeconomic status and higher area‐level deprivation are associated with an increased risk of diabetic ketoacidosis in people with diabetes mellitus type 1.
Previously considered universally fatal, the risk of death with adequate and timely treatment is around 1–4%. Up to 1% of children with DKA develop a complication known as cerebral edema. Rates of cerebral edema in US children with DKA have risen from 0.4% in 2002 to 0.7% in 2012. Between 2 and 5 out of 10 children who develop brain swelling will die as a result.
History
The first full description of diabetic ketoacidosis is attributed to Julius Dreschfeld, a German pathologist working in Manchester, United Kingdom. In his description, which he gave in an 1886 lecture at the Royal College of Physicians in London, he drew on reports by Adolph Kussmaul as well as described the main ketones, acetoacetate and β-hydroxybutyrate, and their chemical determination. The condition remained almost universally fatal until the discovery of insulin in the 1920s; by the 1930s, mortality had fallen to 29 percent, and by the 1950s it had become less than 10 percent. The entity of cerebral edema due to DKA was described in 1936 by a team of doctors from Philadelphia.

RESEARCH STUDIES Numerous research studies since the 1950s have focused on the ideal treatment for diabetic ketoacidosis. A significant proportion of these studies have been conducted at the University of Tennessee Health Science Center and Emory University School of Medicine. Treatment options studied have included high- or low-dose intravenous, subcutaneous, or intramuscular (e.g. the “Alberti regime”) insulin, phosphate supplementation, need for a loading dose of insulin, and appropriateness of using bicarbonate therapy in moderate DKA. Various questions remain unanswered, such as whether bicarbonate administration in severe DKA makes any real difference to the clinical course, and whether an insulin-loading reading reading dose is needed in adults.
The entity of ketosis-prone type 2 diabetes was first fully described in 1987 after several preceding case reports. It was initially thought to be a form of maturity-onset diabetes of the young and went through several other descriptive names (such as “idiopathic type 1 diabetes”, “Flatbush diabetes”, “atypical diabetes” and “type 1.5 diabetes”) before the current terminology of “ketosis-prone type 2 diabetes” was adopted.












Oculus Quest 2 Gelişmiş All in One Sanal Gerçeklik Başlığı 64GB.
Playstation 5 PS5 En Uygun Fiyatla Satın Al.
Oculus Quest 2 Gelişmiş All in One Sanal Gerçeklik Başlığı 256GB.
Oculus Quest 2 Gelişmiş All in One Sanal Gerçeklik Başlığı 256GB.
VR/Sanal Gerçeklik gözlükleri ve aksesuarlarını en uygun fiyatla satın alın.
Oculus Quest 2 Gelişmiş All in One Sanal Gerçeklik Başlığı.
GÜÇLÜ NEODYUM MIKNATISLAR Mıknatıslara gün içerisindeki yapmış olduğumuz birçok alanda ihtiyaç duymaktayız.
Mıknatıslar hayatımıza bir sürü artılar katmaktadır.
Şahsımıza ait hobi amaçlı yaptığımız ürünlerde,
sağlık problemlerimizde kullanmış olduğumuz aletlerimizde ve bunun gibi
birçok alanda mıknatıslara ihtiyaç duymaktayız.
Neodyum mıknatıslar diğer mıknatıslara oranla bu ihtiyaçlarımızın her birini
diğer ürünlere göre bizlere hem estetik açıdan hem yüksek çekim gücü açısından kullanmış olduğumuz alanlarda bizlerin daha fazla verim almamızı sağlamaktadır.
I’m truly enjoying the design and layout of your website.
It’s a very easy on the eyes which makes it much more pleasant for me to come here and visit more often. Did you hire
out a developer to create your theme? Fantastic work!
Yetişkin Sepeti – Türkiye’nin En Güvenilir Cinsel Sağlık Sitesi
hacklink ve Instagram hizmetleri için en doğru adtrestesiniz.
quick credit
web site index.
Facebook & Instagram Reklamcılığı eğitimi ile sizde ileri seviye bir sosyal medya uzmanı olmak için izlemeye başlayın.
Facebook post and status have become a unique way of expressing yourself and even expanding your business.
Web site indexable. Very good web site.
recommended article
faultless article
Tanhu; Toptan ve Perakende, İthalat ve İhracat Seçenekleri ile, İstediğiniz Ürünleri ve Markaları Tedarik Edeceğiniz En Güvenilir Şirket ve Alışveriş Ortamı.!
Tanhu; Toptan ve Perakende Ürün Seçeneklerimiz ile Alışveriş Yapabileceğiniz, Seçenekleri Görebilirsiniz. Gönül Rahatlığıyla Güvenli Alışveriş Yapabilirsiniz.!
Tanhu; İthalat ve İhracat Seçenekleri ile, Toptan ve Perakende, İhtiyacınız Olan Ürünleri Tedarik Edeceğiniz En İyi Şirket ve Güvenilir Alışveriş Ortamı.!Tanhu; Toptan ve Perakende Alışveriş İçin, En Doğru Adres. Gönül Rahatlığıyla Alışveriş Yapabileceğiniz, Yeni Nesil Güvenilir Bir Alışveriş Platformu.!
perfect post, i love it
fake rolex daytona is in pursuit of superior quality.
Global Low-down About this offshoot
https://ivermectinstrom.com ivermectin
Accustomed Information Here this product
https://ivermectinstrom.com buy ivermectin
General Dope Far this by-product
https://ivermectinstrom.com is ivermectin a prescription drug
I want to see you.
replicas hublot watches shoulders the great responsibility of establishment to society.
hublot big bang replica earliest produced a accomplish watch gets results.
best rolex day date replica may really to have a 100 watch business..
fake rolex milgauss has got the planet’s biggest watch innovations.
Every one of fake rolex watch has been given a remarkable watchmaking connotation .
fake rolex has a remarkable modern design.
best rolex cellini replica certainly is the earliest to brew a accomplish watch business..
omega replica primarily developed completed watch goes to work.
BVS – Biblioteca Virtual en Salud – Honduras.
General Low-down Fro this by-product
https://ivermectinst.com ivermectin uk
BVS – Biblioteca Virtual en Salud – Honduras.
BVS – Biblioteca Virtual en Salud – Honduras.
itulkfv wggf70 http://iveramectin.com ivermectin tablets dosage
BVS – Biblioteca Virtual en Salud – Honduras.
Youtube’da ihtiyaçlarınıza uygun paketlerle etkileşimi artırmaya ve hesabınızı geliştirmeye hazır mısınız?
General Information Far this by-product
https://ivermectinsts.com ivermectin uk
Global Information Fro this product
https://ivermectinst.com ivermectin 3mg
General Message Here this offshoot
https://ivermectinsts.com ivermectin for sale
Global Message Far this outcome
https://ivermectinsts.com buy ivermectin
Accustomed Information Here this outcome
https://ivermectinst.com ivermectin without a doctor prescription
General Information About this by-product
https://ivermectinsts.com ivermectin drug
Global Dope About this outcome
https://ivermectinstrom.com where to buy ivermectin
Global Dope Far this product
https://ivermectinstrom.com buy ivermectin online
Non-specific Dope Fro this by-product
https://ivermectinstrom.com where to buy ivermectin for humans
Accustomed Low-down About this product
https://ivermectinst.com ivermectin for sale
Global Low-down Far this outcome
https://ivermectinstrom.com ivermectin tablets cost
Accustomed Dope Here this offshoot
https://ivermectinst.com ivermectin pills for humans
Accustomed Message Far this outcome
https://ivermectinst.com que es la ivermectina
https://ivermectinsts.com ivermectin australia
https://ivermectinst.com ivermectin where to buy
https://ivermectinstrom.com ivermectin otc
https://ivermectstromect.com ivermectin side effects
best thesis thesis editing service
dissertation example dissertation editing service
Global Information Far this outcome
https://ivermectinsts.com ivermectin 3 mg
https://ivermectstromect.com ivermectin human tablets
https://ivermectinst.com ivermectin over the counter
https://ivermectinsts.com ivermectin tablets for humans
https://ivermectinstrom.com ivermectin 6 mg
Non-specific Message Here this by-product
https://ivermectstromect.com ivermectin 3 mg
https://ivermectstromect.com ivermectin for sale
https://ivermectstromect.com ivermectin tablets for humans
https://ivermectinst.com ivermectin for humans
https://ivermectinsts.com ivermectin for humans
ivermectin Ъ©Щ€Ш±Щ€Щ†Ш§ Щ€Ш§Ш¦Ш±Ші https://ivermectinhum.com/ ivermectin tablets
Non-specific Dope Here this offshoot
https://ivermectinstrom.com ivermectin over the counter
https://ivermectinst.com ivermectin 6 mg
https://ivermectinstrom.com ivermectin tablets for humans
https://ivermectstromect.com ivermectin over the counter
https://ivermectinst.com ivermectin 3 mg
Evden eve nakliyat
http://ivermectinovercounter.com – ivermectin pills for humans
ЖЧДF (Lauren)
I ordered the proboscis eradicate, moisturiser, lip balm and soap. Absolutely astonishing consequence barely detail I tend the fuselage collyrium came in a bigger container. Patron handling canadian government approved pharmacies. Blame you
dope vid. the objects section from neighbor has some really nice decor !
So often it seems that you create the article that I need at the time
when I am working on something. And I was really thinking about my wrists lately.
I’ve found that I have to slow down my backswing with the
driver vs itons because the added length makes
it easy to break from how I want to set my wrists at the top.
Is that an indication that something else needs to be
fixed? Thanks for another great article!
Driving a luxury car in Dubai is easier than you might think.
Ferraris, Rolls Royce Phantoms, and other expensive vehicles aren’t
rare to spot in the city. You are able to hire the latest models of those cars at a surprisingly good deal from Luxury Car Rental in UAE.
With an easy telephone call, you are able to rent an automobile with
a high-class interior and a driver who’s acquainted with the area’s traffic rules.
Countless value snow tires. Lenient purchase and had the tires in less than two days. 3rd chance azithromycintok purchaser. Will operation 1010 since any irk needs
So, it appears like a serious part of the plot
will revolve across the sordid past of this residence.
п»їWent to z pack azithromycin where to buy as a replacement for my booster today. Extremely adept and expressively organised, and quite friendly.
alluponurol uses – http://www.inhalerotc.com
You stated this very well.
Hey! Do you use Twitter? I’d like to follow you if that would be okay.
I’m undoubtedly enjoying your blog and look forward to new posts.
Hi there! Do you use Twitter? I’d like to follow you if that would be okay.
I’m definitely enjoying your blog and look forward to new
updates.
My ancestors moved to Cloverdale from Vancouver 23 years ago , and we be subjected to been with pharmasave from the start. I don’t be acquainted with what we would do if we perpetually had to move again , cloverdale pharmasave is like kinsmen , always there when we need you In the gone and forgotten 5 years our sons dog ( Keeda ) has had a totally unfortunate click in her hips that was causing her lots of disquiet, but with the resist of the crozier she just now gets glucosamine everyday and she feels well-advised to it. Thanks hfaventolin.
Wow tons of wonderful tips.
Very good article. I am experiencing some of these issues as well..
Sukces nigdy nie jest ostateczny. Porażka nigdy nie jest totalna.
Liczy się tylko odwaga i waleczne serce. Sir. W. Churchill Adaptation by ITI Systems
Quite insightful submit. Never believed that
it was this simple after all. I had spent a beneficial deal of my time
looking for someone to explain this subject clearly and you’re the
only one that ever did that. Kudos to you! Keep it up
Love it.. keep striving Queen
fildena 100mg reviews fildena 25 mg who sells fildena
Fellow help was deferential and quick responded to my questions ivermectin cream ivermectablets. Coming from another company that wont be named this was a delicious change. Desire be ordering again!
You’ve made your stand pretty effectively!.
Cinemark doesn’t promote personal information of minors under
sixteen years of age with out affirmative authorization.
Palm Springs LA
blogspot.com
Absolutely pent written content, Really enjoyed examining.
Njan purchase pannom pona week super collection.low price
When I originally commented I seem to have clicked on the -Notify me when new comments are
added- checkbox and now every time a comment is added I get four emails
with the exact same comment. Perhaps there is a way you can remove me
from that service? Appreciate it!
This is actually valuable I hope everybody gets the
point youre trying to make here. Im sure all of us readers appreciate
your efforts as much as me!
My family members always say that I am wasting my time here
at net, but I know I am getting experience daily by reading such good articles
or reviews.
Madina Sofa & Chair Upholstery
Opp Mohammad Ali Bin Beyyat Masjid -30 6b St – Al
Barsha
Dubai
0507857283
cost of sofa”
that was perfect
Incredible story there. What happened after? Thanks!
I do not even know how I ended up here, but I thought this post was great.
I don’t know who you are but certainly you’re going to a famous
blogger if you aren’t already 😉 Cheers!
Incredible story there. What occurred after? Thanks!
I set up my protein and creatine that Amazon was into the open of and got the most suitable price bonus accelerated unregulated rid of shipping. Who needs prime at $120 a year. Note Me! Thank you buy ivermectin 6mg, I’ll be back. Regards. Good information.
Touche. Great arguments. Keep up the amazing effort.
hydralazine 50 mg dosage atarax does atarax make you tired
Right on my man!
Wonderful views on that!
I delight working with the baton at fildena 100 usa they in reality punctiliousness about my residents. Very good material, Cheers!
otc atarax hydralazine 50 mg picture atarax uk buy
допог москва
Everyone at this office is outstanding and truly cares about your recovery.
It’s a completely different experience than other treatment centers you’re treated like an adult and with respect, unlike
some other facilities.
PGSLOT.SH เป็น PG SLOT เว็บตรงไม่ผ่านเอเย่นต์ มีสล็อต pg เกมใหม่มากมายมากกว่า 500 เกม
เว็บสล็อต pg แตกง่ายกว่าใคร คุณสามารถรับโบนัสได้ตลอดทุกยอดการเติมเงิน
https://pgslot.sh/
pgslot.sh
I’m not sure why but this site is loading very slow for me.
Is anyone else having this issue or is it a issue on my end?
I’ll check back later on and see if the problem still exists.
Hello! This is my first comment here so I just wanted to
give a quick shout out and say I genuinely enjoy reading through your
posts. Can you suggest any other blogs/websites/forums
that go over the same subjects? Thanks a ton!
For centuries, theologians have been explaining the unknowable in terms of the-not-worth-knowing.
Wonderful views on that!
If you’re reading this Jesus Christ loves you unconditionally and
He will never leave you
1 Nisan 2022 Koç Burcunda Yeniay
Why is it I always really feel like you do?
I’m not sure why but this site is loading very slow for me.
Is anyone else having this issue or is it a problem on my end?
I’ll check back later on and see if the problem still exists.
I was just telling my friend about that.
Your way of telling all in this post is actually fastidious, every
one be capable of easily understand it, Thanks a lot.
That’s very good point
There’s certainly a lot to find out about this topic.
I really like all of the points you made.
Ver y descargar Películas y Series en Latino, Español, Subtitulado e ingles, los
últimos estrenos en la mejor calidad HD sin cortes.
Cuevana Online.
That’s a great point
I was just telling my friend about that.
“im feeling a hole in one here” You should
trust your feelings a bit more mate ❤️ Great shot, straight inn.
Love it..
Hello, I enjoy reading all of your post. I like to write a little comment to support you.
Howdy! I’m at work browsing your blog from my
new iphone! Just wanted to say I love reading through your
blog and look forward to all your posts! Carry on the superb work!
công ty TNHH Việt Thủy Sinh được thành lập and hoạt động bài bản và chuyên nghiệp trong lĩnh vực cách xử lý khoảng không gian.
bên tôi luôn đồng hành cùng với quý khách để cộng phổ biến tay mang
đến chất lượng đỉnh cao mang lại người
đặt hàng.
công ty TNHH Việt Thủy Sinh hiện tất cả đội ngũ kỹ sư công nghệ dày dặn bí quyết,
nhiệt huyết, luôn nghiên cứu vớt biên tập cách
tân công nghệ cung cấp cần thiết của khách hàng.
tôi luôn cập nhật gần như công nghệ tiên tiến bắt đầu nhất trong nước và
thế giới để áp dụng vào cụm cách xử trí khoảng trống đến bạn sử
dụng rộng rãi mang thực tiễn tại
VN.
Phương châm đứng đầu của cửa hàng là: Trao “Chất lượng” nhận “Niềm tin”, chất lượng cao thành
công bắt đầu từ khâu bề ngoài.
I didn’t know that.
I didn’t know that.
Great delivery. Solid arguments. Keep up the
amazing effort.
Miles de Películas y series Online en calidad HD, Castellano y Subtitulado sin cortes.
I didn’t know that.
Pelisplus ⚡ Ver películas online en español latino y series de estreno completas
con todos sus capítulos gratis ⭐ Pelis en HD con el plus de
ver en tu TV.
“This putt is slower than Grayson running a 40-yard dash”
The staff here is awesome, they listen to you, unlike work care
occupational health in our city.
PilarFor Ortho #gennick[XYZ,4,8 #gennick[XYZ,4,8
I didn’t know that.
چرا استعدادیابی و آشنایی با تست استعدادیابی اوکانر مهم
است؟
تست استعدادیابی اوکانر یکی از ابزار
استعدادیابی خصوصا استعدادیابی شغلی است.
ما در این مطلب قصد داریم شما را با این تست
استعدادیابی آشنا کنیم. تا انتهای
مطلب با ما همراه باشید.
آرزوی همه ما این است که بتوانیم در
زندگی موفق شویم. برای موفق شدن در یک زمینه آیتم های مختلفی وجود دارد
که یکی از آنها استعداد است. استعدادها توانایی های بالقوه ما برای انجام امور هستند.
وقتی شما در یک کاری استعداد دارید
سریعتر از دیگران آن را یاد گرفته
و راحت تر انجامش می دهید.
هنگام انجام دادن آن حس خوبی
دارید و پیشرفت سریعی در آن احساس می کنید
طبیعی است که موفقیت در مسیری که شما در آن استعداد
دارید بسیار محتمل تر و راحت تر از مسیری است که
در آن استعداد ندارید. و اینجاست
که اهمیت استعدادیابی مشخص می شود.
شناسایی استعداد شما اگرچه موفقیت شما را تضمین نمی کند اما شانس موفقیت را بسیار
بالا می برد. چرا که شما به جای سرمایه گذاری در مسیری
که ممکن است با مشقت فراوان بتوانید به سطح متوسطی از موفقیت در آن دست یابید مستقیما وارد مسیری می شوید که
شانس موفقیت در آن بالاست.
خب حالا که با اهمیت استعدادیابی آشنا شدید می خواهیم در ادامه به یکی
از ابزارهای استعدادیابی یعنی تست استعدادیابی
اوکانر می پردازیم
معرفی تست استعدادیابی اوکانر
در مقالات قبلی به معرفی تست های گاردنر و هالند برای استعداد یابی
پرداختیم. اکنون می خواهیم تست اوکانر را به شما معرفی کنیم.
اوکانر را باید از پیش گامان
استعدادیابی علمی امروزی نامید.
جانسون اوکانر که در رشته فلسفه از دانشگاه هاوارد فارغ التحصیل شده بود در سال 1922 مسئولیت پروژه آزمون استعدادیابی یا تشخیص
استعداد کارکنان در جنرال الکتریک را
بر عهده گرفت. آن زمان این پروژه با
عنوان مهندسی انسانی نامگذاری شده بود
و در توصیف آن گفته میشد که قرار است هر کس، در شغلی مشغول کار
شود که توانمندی بیشتری برای آن دارد.
تست اوکانر برای این منظور توسط او طراحی و اجرا شد.
مدیران جنرال الکتریک بر این
باور بودند که با این کار، هم کارگران شرایط بهتر و محیط کار
مطلوبتری خواهند داشت و هم خروجی مجموعه بیشتر خواهد شد.
تست استعدادیابی اوکانر
چه مواردی را می سنجد؟
این تست 14 استعداد را در افراد
می سنجد. این استعدادها عبارتند از:
تجسم سازه (structural visualization)
استدلال استقرایی (Inductive Reasoning)
ایده پردازی (Ideaphoria)
سرعت بالای درک دیداری (Graphoria)
استدلال تحلیلی (Analytical Reasoning)
استعدادهای عددی (The Numerical Aptitudes)
استعدادهاس شنیداری (Auditory Aptitudes)
سیلوگرام (Silograms)
حافظه طراحی و مشاهده(Memory for Design & Observation)
تشخیص و ادراک رنگ (Color Discrimination & Color
Perception)
طراحی بصری (Visual Designs)
اشتعدا کار با ابزار و دستان(The Dexterity
Aptitudes)
آینده نگری (Foresight)
ترکیب کلمات (Word Association)
در ادامه هر یک از این موارد را برای شما توضیح خواهیم داد.
استعداد تجسم سازه
افرادی که این استعداد را دارند اغلب جدا
کردن قطعات و کنار هم قرار دادن آنها
را دوست دارند. آنها دوست دارند اشیای جدید طراحی
کرده و آنها را بسازند. این استعداد تجسم سه بعدی شما را نشان می دهد.
این استعداد در تست استعدادیابی
جانسون اوکانر با دو آزمون بلوکه های چوبی بهم ریخته و تا کردن کاغذ سنجیده می شود.
Miles de Películas y series Online en calidad HD, Castellano y Subtitulado
sin cortes.
What is the electric trolley that you use?
I highly recommend them. It’s a great place with excellent facilities and therapy.
The support staff is amazing and wonderful!
Right on my man!
O som tá de boa.
Wonderful views on that!
Can you tell us more about this? I’d want to find out some additional information.
How do I go about copyrighting content of my website?
Tentu aja, dari layanannya ini SLOT88 memberikan minimal
depo yang nilainya amat rendah. Cukup dengan 10ribu, jelas
aja pemain dapat memainkan seluruh permainan slot gacor
yang jumlahnya mencapai ribuan jenis game slot yang nomor
1.Proses depositnya sangat segera. Cukup dengan mentransferkan nilai
depositnya ke akun link aja daripada SLOT88 yang sebelumnya telah Bossku dapatkan dengan menanyakan langsung
pada customer servicenya. Kalau belum mengetahuinya, maka bossque bisa menanyakan langung ke admin dari
romanobet yang aktif terus selama 24 jam pada setiap harinya.
Pastinya Anda sudah familiar dengan nama-nama Penyedia Game slot online terlengkap
seperti Pragmatic play, PG SOFT , HABANERO, SPADE GAMING, TOP TREND
GAMING serta JOKER GAMING. Jenis permainan yang mereka luncurkan paling banyak dimainkan di
dunia. Dengan index keyakinan para penghobi judi slot online tertinggi.
Masing-masing produsen punya puluhan sampai ratusan permainan slot online yang
dapat Bossque mainkan sesuai kesukaan Bossque.
my website slot deposit dana 5000 tanpa potongan 2022
This post was very nicely written :), and it also contains a lot of useful extra facts.
I enjoyed your professional way of writing this post.
Thanks, you have made it very easy for me to understand
Interesing Links in
https://www.alquds.edu/en/
alquds university,جامعة القدس,aqu,arab university,palestinian university
Al-Quds University (AQU) is a collegiate research university that currently offers more than 120 undergraduate and postgraduate programs,
taught through its fifteen degree-granting faculties that cover the main scientific disciplines such as medicine, life, and natural
sciences, business and management, arts and humanities, law and jurisprudence, engineering,
and social sciences. Established in 1970, AQU
remains the only Arab Palestinian university in Jerusalem, Palestine.
AQU currently enrolls around 12,000 full time students,
taught by more than 700 faculty members
I would like to kudos the vastly deferential and welcoming troupe from ivermectin 3 mg tab who without any distrust provided a 5* service. A sportive together, they were dressed smartly and the chemist’s shop was lave and arrange .Above I regard highly the very proper quality conditioning!! Thanks. Loads of tips.
I’m almost fully recovered from my back surgery and could use
a new set
Great article. I will be experiencing a few of these issues as well..
Jestem stara, zostalo mi pewnie malo czasu, nie marnuje go wiec na bzdury.
Robie tylko to, co mnie interesuje. I wam, mlodszym, radze to samo.
– Iris Apfel
ترک خود ارضایی از نظر علمی کاملا ممکن
است. بخش زیادی از ترک اعتیاد به این عمل مانند هرگونه اعتیاد
دیگری جنبه های روحی و روانی زیادی
دارد. بنابر این نیاز است که
با تمام اراده خود تصمیم به ترک گرفته باشید
و از شکست هراسی نداشته باشید.
در ادامه راه هایی برای ترک خود ارضایی که از نظر علمی
ثابت شده اند را بازگو می کنیم.
ورزش کنید
ورزش انرژی مازاد شما را کاهش می دهد.
ورزش علاوه بر آن شادابی و نشاط نیز با خود به ارمغان
می آورد. ورزش هایی مثل دوییدن و شنا می
توانند باعث ترشح اندورفین در بدن می شود.
اندورفین هورمون شادی و نشاط در بدن است.
همچنین ورزش هایی مانند یوگا می تواند آرامش و اضطراب
ناشی از ترک خود ارضایی را به طور چشمگیری کاهش دهد و موجب آرامش هرچه بیشتر گردد.
تغذیه سالم
تغذیه سالم با میوه و سبزیجات کافی می تواند همیشه
سطح انرژی شما را در سطحی متعادل نگه دارد و احساس خستگی
و کسلی را از افراد دور نماید. احساس سرزندگی و نشاط می تواند افراد
را از کارهایی مانند خود ارضایی که که به بدن آسیب وارد
می کند منع نماید.
خواب کافی
کم خوابی با خود بی هدفی و کسالت
به دنبال خواهد آورد. نداشتن
خواب کافی به مرور افراد
را دچار افسردگی می کند. تنظیم عادت خواب و
همچنین خواب کافی به میزان 8 ساعت در روز می تواند احساس افسردگی را از افراد دور می کند.
حمام آب سرد
حمام آب سرد در زمانی که وسوسه های خود ارضایی به سراغ افراد می آید می تواند
معجزه کند. این روش به خوبی می تواند جلوی هر تکرار
عمل خود ارضای را بگیرد.
تنها نباشید
معمولا خودارضایی و وسوسه های آن زمان به
سراغ افراد می آید که تنها هستند.
به همین دلیل توصیه می شود برای ترک خود
ارضایی بهتر است خود را در
موقعیت هایی قرار دهید که تنها نباشید.
از تماشای فیلم ها و عکس های مستهجن دوری کنید
فیلم ها و عکس های مستهجن می توانند یک عامل
مهم در شروع وسوسه برای خود ارضایی باشد.
ترک خود ارضایی زمانی ممکن می شود که
افراد عامل محرک وسوسه ها را پاک کنند.
فیلم های پورن و عکس های مربوط به صحنه
های سکسی به طور یقین یکی از همین
عوامل محرک است که افراد به راحتی می توانند
از دیدن آن صرف نظر کنند.( اختلال تماشاگری
جنسی)
از یک مشاور کمک بگیرید
Egal ob jemand sein notwendiges Ding sucht, daher er/sie will, dass im Detail verfügbar sein, so
wird das Ding hier beibehalten.
Si tienes ganas de tener decoración salón comedor rectangular mira este catálogo barato
Great items from you, man. I have keep in mind your stuff previous to and you
are just extremely excellent. I actually like what you have obtained right here, really like
what you are stating and the way in which in which you are saying it.
You’re making it entertaining and you still take care of to stay it smart.
I cant wait to learn far more from you. That is actually a terrific
website.
m health maple grove and r news anchors and health belief model and news now and p health pillow and fox news live
and rstudio education and b education primary uon
RDVX CVZB 4ADW DJP3 2ADQ 4VWG MNAM KIU6
I am truly thankful to the holder of this site who has shared this great
post at at this time.
It’s impressive that you are getting ideas from this piece of
writing as well as from our dialogue made at this place.
Appreciate the recommendation. Let me try it out.
If you wish for to improve your knowledge only keep visiting this
web site and be updated with the latest news update posted here.
Karenanya, prioritaskan website yang punyai penampilan bagus.
Tidak dapat disangkal kalau website yang penampilannya bagus itu betul-benar dibutuhkan. Mata bakal benar – betul dimanja dengan tampila website yang bagus dan mengagumkan cantiknya.
Begitu jejeran persyaratan dari sebuah website judi slot pulsa paling dipercaya dan sah.
Dengan menyaksikan persyaratan – persyaratan di atas, seluruh keringanan di waktu
menentukan sebuah website judi slo deposit pulsa bakalan benar – benar
didapat oleh beberapa playernya.
How to get your PMP certification?
You need to fulfil the PMP Certification requirements.
Review the PMP exam objectives: review the PMP Examination Content Outline
(ECO) to make sure you know what is covered in the exam.
You will need to take a training course to obtain your
35 hours of contact hour.
Practice for the exam: Practice, Practice, Practice, after you have studied for the exam, review,
and answer as many practice exams as you can. Check the free PMP Practice
Exams , and the Premium Practice exams
Submit your application.
Take the test: Once your application is approved, you will be given information on payment and scheduling options.
That’s very good point
Many sources should really be utilized in order to make
an informed lawwyer choiche selection.
My website 여성알바사이트
Hey there! Do you use Twitter? I’d like to follow you if that would be okay.
I’m absolutely enjoying your blog and look forward to new updates.
Right on my man!
Thanks in support of sharing such a nice idea, post is pleasant, thats
why i have read it entirely
I’m truly enjoying the design and layout of your site.
It’s a very easy on the eyes which makes it much
more pleasant for me to come here and visit more often. Did
you hire out a designer to create your theme? Fantastic work!
linear algebra and its applications 6th edition pdf
BestFootballTips hit tomorrow soccer prediction from whiz.
footyaccumulators
Best Football Tips com it’s a easy to understand Web page which
offers free betting tips soccer. oddschecker
BFT it’s a handy database with miscellaneous functionalities.
BestFootballTips.com take advice to Best Football Tips com
customer criticism and positioned on that we have donate
a undoubtedly orderly network BFT. footyaccumulators
Best Football Tips com feeler an commodious range of unpaid and reimbursed super tips bet along with alive issue and last news,
deportation, pre match best part. sportsmole
Really good info can be found on blog.
I really like looking through a post that will make men and
women think. Also, many thanks for allowing me
to comment!
ترک خود ارضایی از نظر علمی کاملا ممکن است.
بخش زیادی از ترک اعتیاد
به این عمل مانند هرگونه اعتیاد دیگری جنبه های
روحی و روانی زیادی دارد. بنابر این نیاز است
که با تمام اراده خود تصمیم به ترک گرفته
باشید و از شکست هراسی نداشته باشید.
در ادامه راه هایی برای ترک
خود ارضایی که از نظر علمی
ثابت شده اند را بازگو می کنیم.
ورزش کنید
ورزش انرژی مازاد شما را کاهش می دهد.
ورزش علاوه بر آن شادابی و نشاط نیز با خود به ارمغان می آورد.
ورزش هایی مثل دوییدن و شنا می توانند
باعث ترشح اندورفین در بدن می شود.
اندورفین هورمون شادی و نشاط در بدن است.
همچنین ورزش هایی مانند یوگا می تواند آرامش و اضطراب ناشی از ترک خود ارضایی را به طور
چشمگیری کاهش دهد و موجب آرامش هرچه بیشتر گردد.
تغذیه سالم
تغذیه سالم با میوه و سبزیجات کافی می تواند همیشه سطح انرژی شما را در سطحی متعادل نگه دارد و احساس خستگی
و کسلی را از افراد دور نماید. احساس سرزندگی و نشاط می تواند افراد را
از کارهایی مانند خود ارضایی که که به بدن آسیب وارد می کند منع نماید.
خواب کافی
کم خوابی با خود بی هدفی و کسالت به
دنبال خواهد آورد. نداشتن خواب کافی
به مرور افراد را دچار افسردگی می کند.
تنظیم عادت خواب و همچنین خواب کافی به میزان 8 ساعت در روز می تواند احساس افسردگی را از افراد دور می کند.
حمام آب سرد
حمام آب سرد در زمانی که وسوسه های خود ارضایی به سراغ افراد می
آید می تواند معجزه کند. این روش
به خوبی می تواند جلوی هر تکرار عمل خود ارضای را بگیرد.
تنها نباشید
معمولا خودارضایی و وسوسه های آن زمان به
سراغ افراد می آید که تنها هستند.
به همین دلیل توصیه می شود برای ترک خود ارضایی بهتر است خود را در موقعیت هایی قرار دهید که تنها نباشید.
از تماشای فیلم ها و عکس های مستهجن دوری
کنید
فیلم ها و عکس های مستهجن می توانند یک عامل مهم در شروع
وسوسه برای خود ارضایی باشد. ترک خود ارضایی زمانی ممکن می شود که افراد عامل محرک وسوسه ها را پاک کنند.
فیلم های پورن و عکس های مربوط به صحنه های سکسی به طور یقین یکی از همین عوامل محرک
است که افراد به راحتی می توانند از دیدن آن صرف نظر کنند.( اختلال تماشاگری جنسی)
از یک مشاور کمک بگیرید
The Northman is an epic revenge thriller, that explores how far a
Viking prince will go to seek justice for his murdered father.
بروس وین در اوایل فیلم بتمن (The Batman)
می خواهد به ما بگوید که ترس
یک ابزار است. او در این مورد صحبت می کند که چگونه می توان از حضور بتمن
برای ترساندن افراد بد استفاده کرد، اما ممکن است مت ریوز (نویسنده/کارگردان)،
این موضوع را به خاطر رویکرد خود برای راهاندازی مجدد ابرقهرمان معروف مورد توجه قرار
دهد. این فیلم، ترسناک ترین بتمن است.
درست از صحنه آغازین خشونت آمیز، پیام واضح است:
این کاپیتان صلیبی مادر شما نیست.
این فیلم، یک فیلم هیجانانگیز روانشناختی، خشم آلود و مهیج با دوز
سنگینی از صحنه های جنایی است و
باور کنید یا نه، ریوز کاملاً هنری این صحنه ها را
اجرا می کند و به شاهکاری خیلی زیبا دست مییابد.
بتمن سر جای خود ایستاده است، اما همچنان مملو از ارجاعات سینمایی است.
اکثر فیلم های قبلی بتمن لایو اکشن است.
رئالیسم سنگدل موجود در این فیلم بیشتر شبیه به فیلم سه
گانه کریستوفر نولان است، اما این یک برداشت سینمایی تازه و جسورانه از شوالیه
تاریکی (Dark Knight) است.
نقد فیلم بتمن The Batman
نقد فیلم بتمن The Batman
در هر صورت، ماهیت پایه این فیلم خیلی شبیه به جوکر 2019 است.
اما تفاوت اینجاست که تریلر خواکین فینیکس واقعاً نیازی
به نام شخصیت شرور نداشت تا داستان مردی فقیر را که توسط جامعه فراموش شده است را روایت کند.
از سوی دیگر، بتمن، هنوز هم به طرز شگفتآوری وفادار داستان بتمن است.
این داستان از داستانه ای مختلفی از کمیکها به شیوهای
متهورانه و در عین حال محترمانه میکس می کند،
در حالی که همه اینها با آنچه تا به اینجا در
صفحه بزرگ سینما دیدهایم خیلی
متفاوت است.
فقط به یک دلیل منشأ این داستان بتمن نیست.
ریوز میداند که ما می دانیم توماس و مارتا وین مردهاند
و به درستی فرض می کند که دیگر نیازی به تیراندازی
به آنها نداریم. در عوض، ما مستقیماً وارد شراکت بتمن و
کارآگاهی بتمن و جیم گوردون شده ایم.
این موضوع آنقدر در داستان بروس
وین اتفاق می افتد که صحنههایی را که قبلاً میلیون ها بار دیدهایم بازخوانی نمی کند،
در واقع او هنوز باید کارهای زیادی انجام دهد تا اینکه ابرقهرمان
تقریباً بیعیب و نقصی خلق کند.
ما آغاز را نمی بینیم، اما شاهد پیشرفت
های زیادی هستیم و همچنین برخی
از فراخوان های هوشمندانه و اضافه شده به تاریخچه چندین
خانواده گاتهام را می بینیم.
Right on my man!
terima kasih telah berbagi berita Pak, senang sekali rasanya membaca tulisan2 di blog ini, saya merasa bertambah wawasan semoga selalu di beri kesehatan dan sukses selalu..
I’m really enjoying the design and layout of your blog.
It’s a very easy on the eyes which makes it much more
enjoyable for me to come here and visit more often. Did
you hire out a designer to create your theme?
Fantastic work!
قیمت سئوی سایت و بهینه سازی سایت جوملا و تولید محتوا علوم پنجم و فتوشاپ در طراحی سایت
و سایت های بهینه سازی عکس و
طراحی سایت تجارت الکترونیک
https://majalehirani.ir/2022/آشنایی-با-شرایط-دریافت-خدمات-مشاوره-سئ/
This is the perfect site for anyone who hopes to understand
this topic. You realize so much its almost hard to argue
with you (not that I really will need to…HaHa).
You certainly put a new spin on a subject which has been discussed
for ages. Wonderful stuff, just wonderful!
El desfile finalizó con la liberación al cielo de decenas de miles de globos de colores, realizada por cientos de niños, mientras que en el fondo se
escuchaba la consigna “Viva la República Popular China”.
Podemos hablar hasta de centros de mesas creados con globos y es que los globos le
dan alegría a una fiesta. Podemos personalizarte desde una decoración insólita para equipar tu
escritorio, hasta un accesorio de marroquinería que te acompañe a todas tus citas o un cojín personalizado con la foto de tu perro
o tu bonita familia, ¡ Podemos fabricarlos además en cartón o en noble madera impresa.
También puedes adquirir lienzos personalizados (sin bastidor), enmarcaciones en aluminio o madera, pósters personalizados, fotos en madera,
fotos en metacrilato, fotos en aluminio, fotos en cartón pluma, lonas publicitarias y otros productos originales como estores personalizados, placas de metacrilato, biombos personalizados, roll ups y otros productos para decoración. Confeccionamos
todo tipo de regalos, con una enorme variedad que alcanza hasta unos 500 productos diferentes, a partir de las fotos y textos que nos faciliten nuestros
clientes. Sus piezas realistas y minifiguras, entre las
que encontrarás al insustituible Luke Skywalker, trasladará a
todos los jugadores a las batallas galácticas más importantes.
I’d like to find out more? I’d like to find out more details.
Hey there! Do you use Twitter? I’d like to follow you if that would
be okay. I’m definitely enjoying your blog and look forward to new updates.
chloroquine chloroquine
สล็อตเว็บตรง สล็อต PG ไม่ผ่านเอเย่นต์
สมัครสล็อตpg ทดลองเล่นได้แล้วที่นี่ PGTHAI.CLUB
สมัคร สล็อต pg แตกง่าย
pgthai
I’m really enjoying the design and layout of your website.
It’s a very easy on the eyes which makes it much more pleasant
for me to come here and visit more often. Did you hire out a designer to create your theme?
Outstanding work!
https://pharmduck.com/ over the counter drugs
She’s already cried once this morning about what won’t be right here next month.
Hi! Do you use Twitter? I’d like to follow you if that would be
okay. I’m definitely enjoying your blog and look forward
to new updates.
This info is invaluable. Where can I find out more?
I’m truly enjoying the design and layout of your website.
It’s a very easy on the eyes which makes it much more pleasant for me to
come here and visit more often. Did you hire out a designer to create your theme?
Outstanding work!
I’m not sure exactly why but this site is loading very slow for me.
Is anyone else having this issue or is it a problem on my end?
I’ll check back later on and see if the problem still exists.
Really, this is a handy site.
https://clubswitch.co.za/author/alessandrahennesseyhd/
At this time I am going to do my breakfast, when having my breakfast
coming yet again to read other news.
I felt nothing before getting to what I am today. Thank you so much for the experience I’m having.
The staff is very supported at everything and willing to assist in my recovery.
Best I’ve ever been to so far. The staff is very nice and helpful too.
We all deserve to be treated well you know.
This is the perfect site for anyone who wishes to find
out about this topic. You know so much its almost hard to argue with
you (not that I really will need to…HaHa).
You definitely put a fresh spin on a subject that’s been written about
for decades. Excellent stuff, just great!
Why is it I always really feel like you do?
I didn’t know that.
สล็อตเว็บตรง สล็อต
PG ไม่ผ่านเอเย่นต์
สมัครสล็อตpg ทดลองเล่นได้แล้วที่นี่ PGTHAI.CLUB
สมัคร สล็อต ppg แตกง่าย
รวมสล็อตpg
Menemukan web terpercaya adalah kunci bagaimana kita dapat mulai bermain dengan gampang di laman hal yang demikian. Berhubungan kita sudah menemukan atau mengontrol kunci hal yang demikian, karenanya metode main taruhan online paling gampang yang dibagikan di
sini akan berguna bagi tiap-tiap pemain yang baru pertama kali bermain di sebuah website betting.
kita juga sepatutnya mengamati sebagian hal lainnya.
Setidaknya kita sepatutnya mengenal beberapa undang-undang dan metode main dari sebuah
permainan sebelum kita mulai bertaruh. Tutorial memudahkan kita dalam memainkan permainan tersebut, kita juga dapat menghindari kesalahan yang berujung pada kerugian kita sebab keok taruhan atau kesalahan pada salah satu jenjang bermain di web judi tertentu.
Howdy! Do you use Twitter? I’d like to follow you if that would be okay.
I’m undoubtedly enjoying your blog and look forward to new posts.
Great beat ! I would like to apprentice while you amend your site, how could i subscribe for a blog
website? The account helped me a acceptable deal. I had been a little bit acquainted of this your broadcast provided bright clear concept
health psychology
http://panjerehmag.tebyan.net/newindex.aspx
I’m really enjoying the design and layout of your website.
It’s a very easy on the eyes which makes it much more pleasant for me to come here and visit more often.
Did you hire out a designer to create your theme?
Fantastic work!
สมัครSLOTXO เว็บสล็อต SLOTXOTH
สมัครสล็อตXO รับโบนัสฟรีที่ SLOTXOTH
SLOTXOTH
slotxo สมัคร
Right on my man!
There is certainly a great deal to learn about this subject.
I love all of the points you made.
PG SLOT เว็บตรง ไม่มีขั้นต่ำ
สล็อตเว็บตรง ไม่ผ่านเอเย่นต์
SLOT PG เว็บใหม่ 2021
slot pg เว็บตรง
http://anonymto.com/?https://sessizlimanyuregim.blogspot.com/ sesizliman yüreklilier blogu
Эта сообщение поможет Интернету пользователям для настройки
нового сети site или даже blog от начала до конца.
двери
It’s impressive that you are getting ideas from this article as well as from our argument made at this place.
May I simply say what a relief to uncover someone who actually understands what they’re talking
about online. You actually realize how to bring an issue to light and make it important.
More and more people should look at this and understand
this side of the story. It’s surprising you are not more popular since you definitely
have the gift.
The official Casino guide to 2022
thailand
ks kurve หรือ kardinal kurve เป็นหนึ่งใน บุหรี่ไฟฟ้า ประเภท pod system ของทางแบรนด์ kardinal โดย
ks kurve นั้น เป็นผลิตภัณฑ์รุ่นใหม่ล่าสุด ที่ได้มีการปรับปรุงมาจากรุ่น kardinal stick ที่ได้มีการปรับเปลี่ยนใหม่ทั้งหมดตั้งแต่ตัวเครื่อง
ks kurve หรือ kardinal kurve ไปจนถึง น้ำยา ks kurve pod หรือ kardinal kurve pod เพื่อแก้ปัญหาที่พบเจอจากรุ่น kardinal stick และเพิ่มประสิทธิภาพในการช่วย เลิกบุหรี่
และตอบโจทย์ทุกการใช้งานของผู้ใช้มากยิ่งขึ้น ด้้วยเทคโนโลยีรุ่นใหม่ล่าสุด ต้องบอกได้เลยว่า
ks kurve หรือ kardinal kurve รุ่นนี้ ทางเจ้าของแบรนด์ kardinal จัดเต็ฒทุกการทำงาน ไม่ว่าจะเป็น ดีไซน์ที่ทันสมัยกว่าเดิม ระบบการสูบที่ดียิ่งขึ้น วัสดุที่ทนทานมากขึ้น ปรับเพิ่มโครงสร้างต่างๆ เพื่อลดปัญหาจากการใช้งาน และที่สำคัญยังมีการเพิ่มประสิทธิภาพในการชาร์จที่เต็มไวกว่าเดิม
เพียง 18 นาทีเท่านั้น ks kurve หรือ kardinal kurve หรือ ks kurve lite
รุ่นนี้ครบจบในเครื่องเดียวจริงๆ
great put up, very informative. I ponder
why the other experts of this sector don’t realize this.
You must continue your writing. I am confident, you’ve a huge readers’ base already!
อย่าห้าวให้มากไอ้เด็กเหี้ย
Citizenship as well as Immigration Service (“USCIS”), requires expertise of the seeking company’s item, solution, study, devices,
techniques, monitoring, or various other interests.
I was just telling my friend about that.
Shelby do you want us to watch your articles?
because your sign says hello but your hoodie says goodbye
Incredible story there. What occurred after? Take care!
The official Cassino guide to 2022
casino
Good article. I am going through some of these issues as well..
อย่าห้าวให้มากไอ้เด็กเหี้ย
Really This goes far beyond the commenting! It wrote his thoughts while reading the article
amazingly 🙂
Minimal Deposit & Withdraw Di Situs Pragmatic189
Kami selaku situs online pragmatic189 dengan rasa hormat yang tinggi mengajak para penjudi
Indonesia untuk bergabung bersama kami cita membangun dan meningkatkan perekonomian. Mulai dari pendaftaran gratis
dan minimal deposit Rp 20.000,- maka penjudi sudah siap mencari nafkah melalui
semua game yang tersedia. Withdraw minimal Rp 50.000.- sampai ratusan juta siap didapati bagi semua pemain setia
pragmatic189. Cukup dengan mencantumkan data diri asli dan aktif maka player sudah
siap dan berhak mendapatkan username dan password untuk selanjutnya
login kedalam akun yang tersedia banyak permainan favorit
dan disukai.
I’d like to find out more? I’d want to find out more details.
Very descriptive article, I enjoyed that a lot. Will there be a part 2?
This information is worth everyone’s attention. Where can I
find out more?
Good web site you have got here.. It’s hard to find
excellent writing like yours these days. I honestly appreciate people
like you! Take care!!
Naprawdę podoba mi się twój szablon wp zwłaszcza ostylowanie CSS,
skąd go pobrałeś? Z góry dziękuję!
HTX Nam Tây Nguyên
Website: Viencaytrong.com và VuaCay.Com
Số điện thoại: 0914599143
Cơ sở cung cấp giống cây ăn trái, cây công nghiệp và
cây lâm nghiệp.
อย่าห้าวให้มากไอ้เด็กเหี้ย
HTX Nam Tây Nguyên
Website: Viencaytrong.com và VuaCay.Com
Số điện thoại: 0914599143
Cơ sở cung cấp giống cây ăn trái,
cây công nghiệp và cây lâm nghiệp.
Howdy! Do you use Twitter? I’d like to follow you
if that would be okay. I’m absolutely enjoying your blog and look forward
to new updates.
Freuen Sie sich hier sommers wie winters auf viele
Möglichkeiten für Wanderungen und Skitouren im winter.
Weitere Ideen zum Urlaub mit Hund in der Schweiz finden Sie hier.
Wie wäre es mit einem NOVASOL Ferienhaus im tschechischen Riesengebirge?
Suchen Sie noch ein günstiges Ziel für Ihren nächsten Urlaub in den Bergen mit Hund?
Für alle wanderbegeisterten Zwei- und Vierbeiner bietet
sich eine Landschaft voll kahler Bergkämme, tiefer Täler,
kristallklarer Bäche und Wasserfälle. Das Riesengebirge erreicht neben anderen das Schneekoppe eine Höhe von 1.603 Metern und zählt zu den beliebtesten Urlaubsregionen in Tschechien. Zufolge Jahreszeit können Sie und Ihr Vierbeiner
auch auf die Suche nach schmackhaften Beeren und Pilzen gehen. Das Eingangstor
ins Riesengebirge stellt die Stadt Turnov dar.
Hier finden Sie die Tradition der Edelsteinbearbeitung und Schmuckherstellung.
Weitere Infos zum Urlaub mit Hund in Tschechien lesen Sie
hier. Entdecken Sie auch die Kunst der Glasbläserei in vielen Orten Tschechiens.
Ein Urlaub in den Bergen mit Hund lässt Sie und Ihren Vierbeiner jeglichen Alltagsstress vergessen und empfängt Sie mit einer
malerischen Naturlandschaft. Unsere Ferienwohnungen in der Hohen Tatra sind hierfür vorzüglich geeignet: traumhafter Panoramablick auf die Bergkulisse, ein eigener Kamin für gemütliche Stunden und ein eingezäuntes Grundstück
für glückliche Vierbeiner.
It is designed with IM7 graphite which is the induce of its lightweight and energy.
In truth, it is like a fusion of strength and light-weight.
So, you see- you get the ease and comfort just the way you like it.
In this section of our best spinning rod for evaluate we’d like to share some spinning rod setup methods.
Apparently, you need to have the rod to be ultralight.
Apparently, the line fat capability is 8-17
lbs . which is pretty a fantastic just one for trout fishing.
Evidently, you will commonly find just one form of tackle popular
with trout spinning rods- split handles. The cause is its manage.
A different factor you have to take into
account is the material of the cope with. This is why you have to fairly watchful when you
are fishing trout. Excess weight is really a pretty significant thing to consider when it comes to trout fishing.
This feature would make it a person of the very best spinning rod for trout out there.
You can check them out as well. The line pounds potential is not
specified for the rods so you will have to find a way to figure
it all out. Mainly because trout are spooked so easily, you have to conceal your existence in any way you can.
Hi there, I enjoy reading all of your post. I wanted to write
a little comment to support you.
Great blog! Is your theme custom made or did you download it from
somewhere? A design like yours with a few simple adjustements would really make my
blog jump out. Please let me know where you got your theme.
Many thanks
PG SLOT เว็บตรง ไม่มีขั้นต่ำ
สล็อตเว็บตรง ไม่ผ่านเอเย่นต์
SLOT PG เว็บใหม่ 2021
สล็อตเว็บตรง
Howdy! Do you use Twitter? I’d like to follow you if that would be
ok. I’m absolutely enjoying your blog and look forward to new
updates.
سرم طلا هیالورونیک اسید 24k بیوآکوا
If you would like to grow your familiarity just keep visiting this
website and be updated with the hottest news posted here.
Very good article. I will be facing many of these issues as well..
ترک خود ارضایی از نظر علمی کاملا ممکن است.
بخش زیادی از ترک اعتیاد به این
عمل مانند هرگونه اعتیاد دیگری جنبه های روحی و روانی زیادی دارد.
بنابر این نیاز است که با تمام اراده
خود تصمیم به ترک گرفته باشید و از شکست هراسی نداشته باشید.
در ادامه راه هایی برای ترک خود ارضایی که
از نظر علمی ثابت شده اند را بازگو می کنیم.
ورزش کنید
ورزش انرژی مازاد شما را کاهش می دهد.
ورزش علاوه بر آن شادابی و نشاط نیز با خود به ارمغان می آورد.
ورزش هایی مثل دوییدن و شنا می توانند باعث ترشح اندورفین در بدن می شود.
اندورفین هورمون شادی و نشاط در بدن
است. همچنین ورزش هایی مانند یوگا می تواند آرامش
و اضطراب ناشی از ترک خود ارضایی را به
طور چشمگیری کاهش دهد و موجب آرامش هرچه بیشتر گردد.
تغذیه سالم
تغذیه سالم با میوه و سبزیجات کافی می تواند همیشه سطح انرژی شما را در سطحی متعادل نگه دارد و احساس خستگی و کسلی را از افراد
دور نماید. احساس سرزندگی و نشاط می تواند
افراد را از کارهایی مانند خود ارضایی
که که به بدن آسیب وارد می کند منع نماید.
خواب کافی
کم خوابی با خود بی هدفی و کسالت به
دنبال خواهد آورد. نداشتن خواب کافی به مرور افراد
را دچار افسردگی می کند. تنظیم عادت خواب و همچنین خواب کافی به
میزان 8 ساعت در روز می تواند احساس افسردگی را
از افراد دور می کند.
حمام آب سرد
حمام آب سرد در زمانی که وسوسه
های خود ارضایی به سراغ افراد می آید
می تواند معجزه کند. این روش به خوبی می تواند جلوی هر تکرار عمل خود ارضای را بگیرد.
تنها نباشید
معمولا خودارضایی و وسوسه
های آن زمان به سراغ افراد می آید
که تنها هستند. به همین دلیل توصیه می شود برای ترک خود ارضایی بهتر است خود را در موقعیت
هایی قرار دهید که تنها نباشید.
از تماشای فیلم ها و عکس های مستهجن دوری
کنید
فیلم ها و عکس های مستهجن می توانند یک عامل مهم
در شروع وسوسه برای خود ارضایی باشد.
ترک خود ارضایی زمانی ممکن می شود که افراد عامل محرک
وسوسه ها را پاک کنند. فیلم های پورن و عکس های مربوط به صحنه
های سکسی به طور یقین یکی از همین عوامل محرک است که افراد
به راحتی می توانند از دیدن آن صرف نظر کنند.( اختلال تماشاگری جنسی)
از یک مشاور کمک بگیرید
Психология отношений между мужчиной и женщиной – это сфера изучения взаимодействия полов на всех уровнях коммуникации.
Проблемы здорового общения, совместимости, конфликтов
и расставания. Механизмы психики,
влияющие на наше поведение в союзе с партнером, для многих кажутся непонятными.
อย่าห้าวให้มากไอ้เด็กเหี้ย
https://beydoun-lb.com/curtains-lebanon-reputation/
curtains lebanon
Mempertimbangkan kalian tertarik untuk bermain dan mencoba peruntungan kalian di
judi slot online, kalian mesti mendaftar dan mempunyai akun untuk
bermain. Jikalau banyak website judi yang ada di internet yang bisa kalian gunakan untuk
bermain judi slot online ini. Metode semacam itu
kalian patut memilih web yang ideal dan juga dapat dipercaya untuk bermain.
Karena tak segala situs yang beredar di dunia online ialah website orisinil.
Menetapkan hingga anda salah memilih situs untuk bermain judi
slot ini, karenanya akan benar-benar disayangkan jumlah uang yang anda telah
gunakan. Pilihlah dengan bijak mengenai laman yang akan kalian pakai
untuk bermain. Carilah isu soal web-situs hal yang demikian di internet dan beraneka sumber lain.
Magnificent items from you, man. I have remember your stuff previous to and you’re just too fantastic.
I really like what you have got here, really like what you
are stating and the best way during which you say it.
You are making it entertaining and you still care for to keep it wise.
I can not wait to read much more from you. That is actually
a terrific web site.
Fine way of explaining, and good article to take data concerning my presentation subject matter,
which i am going to convey in university.
I’m really enjoying the design and layout
of your blog. It’s a very easy on the eyes which makes it
much more pleasant for me to come here and visit more
often. Did you hire out a designer to create your theme?
Excellent work!
Appreciate the recommendation. Let me try it out.
I was just telling my friend about that.
Hello, I enjoy reading all of your article. I wanted to write a little comment
to support you.
I’ve been browsing on-line more than three hours these days, yet
I never found any attention-grabbing article like yours.
It’s beautiful price sufficient for me. In my opinion, if all site
owners and bloggers made good content as you did, the net might be much more helpful than ever before.
อย่าห้าวให้มากไอ้เด็กเหี้ย
PG SLOT เว็บตรง ไม่มีขั้นต่ำ
สล็อตเว็บตรง ไม่ผ่านเอเย่นต์
SLOT PG เว็บใหม่ 2021
pg slot game
Neden Instagram?
Neden herkes bu fotoğraf paylaşım uygulamasıyla ünlü olmak istiyor?
Birçok etkileyicinin ve işletmenin bu kanalı para kazanmak ve bundan büyük miktarda para kazanmak
için kullandığını biliyor musunuz? Bu tanıtıcı yaklaşık
2B’ye ve belki daha fazla hayran kitlesine sahiptir ve aşağıdakiler
gibi çeşitli nişlerden insanlardan oluşur:
moda
elektronik
cilt bakımı
Kullanıcıların her biri için olanaklara sahip olan uçsuz bucaksız okyanustur.
Ancak tek ihtiyacınız olan güvenilir bir plan ve şemadır.
Birçok işletme, sosyal medya işlerini yönetmek için dijital pazarlama uzmanları bile tutar.
Gönderinin yapılmasından takipçi numaralarına kadar tüm konularla ilgilenirler.
Ama kayda değer bir maliyeti var. Herhangi biri öğrenciyse ve bütçesi kısıtlıysa, bu sizin için uygun bir seçim değildir.
Ama unutmayın, şöhret de yatırım gerektirir.
İster sektörde yeni olun, ister eski, bazen markalaşma planınızı beslemeniz gerekir.
Bu yüzden oyun için biraz bükülerek doğru planı alın.
Instagram küçük işletmelerle ünlü olduğundan, çoğu
durumda bir dijital pazarlama ekibi kiralamak bir seçenek değildir.
Ve yeni başlayanların büyümesi için, bu tutamaç, küçük işletme
çıkartmalarını desteklemek gibi çeşitli unsurları devre dışı bıraktı.
o yüzden instagram takipçi satın al
https://cehennemcom.blogspot.com
Among them is Lieutenant Bradley “Rooster” Bradshaw, the son of Maverick’s late finest friend Nick “Goose” Bradshaw.
Instead, Jennifer Connelly steps in as a San Diego bar owner who’s had an on-again, off-again thing with Maverick for decades.
Can you tell us more about this? I’d like to find
out more details.
This site certainly has all oof the info I wanted about this
subject and didn’t know who to ask.
건마
نصب سیستم عامل لینوکس در کنار ویندوز از
دغدغه هایی است که هر شخصی بخطر درگیر شدن درایو مورد نظر جهت نصب لینوکس خودداری میکند؛ به همین منظور، در دسته مبحث آموزش لینوکس،
روش نصب لینوکس در کنار ویندوز در یک درایو را بصورت کامل و دقیق توضیح خواهیم داد…
Why is it I always really feel like you do?
Awesome insight!!!
So who outlasted the rest and who did the jury vote as the winner of
Movie star Big Brother Season 3? Corey Brooks was put on the block, by Roadkill winner Tiffany, however he received the facility of Veto.
He additionally provides that he never sat on the nomination block, and that he needs
to use the money to make artwork for his neighborhood. The cast
members of Big Jake have been in many other movies, so use this
checklist as a starting point to find actors or actresses that you simply will
not be accustomed to. Beginning to make use of a harness
for exercising giant energetic canine may appear exhausting to do but some newer harnesses feature basic however nicely designed techniques just
like the front-attachment fastening factors. It simply may be the Billy
Redden cameo (most famously known as the dueling banjo kid in Deliverance),
however the city reeks of an oncoming Hansel & Gretel style entice.
In case that is the primary time that you’re opting to go for these designers,
the reality is that you would possibly get quite confused.
It comes time for Miesha to evict the final houseguest,
and of course, she boots Cynthia, opting to take Todrick to the finals to face the jury.
I didn’t know that.
อย่าห้าวให้มากไอ้เด็กเหี้ย
ترک خود ارضایی از نظر علمی کاملا ممکن است.
بخش زیادی از ترک اعتیاد به این عمل مانند
هرگونه اعتیاد دیگری جنبه های روحی و روانی زیادی
دارد. بنابر این نیاز است که با تمام اراده خود تصمیم به ترک
گرفته باشید و از شکست هراسی نداشته باشید.
در ادامه راه هایی برای ترک خود ارضایی که از نظر علمی ثابت شده اند را بازگو می کنیم.
ورزش کنید
ورزش انرژی مازاد شما را کاهش می دهد.
ورزش علاوه بر آن شادابی و نشاط نیز با خود به ارمغان می آورد.
ورزش هایی مثل دوییدن و شنا می توانند باعث ترشح اندورفین در بدن می شود.
اندورفین هورمون شادی و نشاط در بدن است.
همچنین ورزش هایی مانند یوگا می تواند آرامش و اضطراب ناشی
از ترک خود ارضایی را به طور چشمگیری کاهش دهد و موجب آرامش هرچه
بیشتر گردد.
تغذیه سالم
تغذیه سالم با میوه و سبزیجات کافی
می تواند همیشه سطح انرژی شما را در سطحی
متعادل نگه دارد و احساس خستگی و کسلی
را از افراد دور نماید. احساس سرزندگی و نشاط می تواند افراد
را از کارهایی مانند خود ارضایی که که
به بدن آسیب وارد می کند منع نماید.
خواب کافی
کم خوابی با خود بی هدفی و کسالت به دنبال خواهد آورد.
نداشتن خواب کافی به مرور افراد
را دچار افسردگی می کند.
تنظیم عادت خواب و همچنین خواب کافی به میزان 8
ساعت در روز می تواند احساس افسردگی
را از افراد دور می کند.
حمام آب سرد
حمام آب سرد در زمانی که وسوسه های
خود ارضایی به سراغ افراد می آید می تواند معجزه
کند. این روش به خوبی می تواند جلوی هر تکرار عمل خود ارضای را بگیرد.
تنها نباشید
معمولا خودارضایی و وسوسه های آن زمان به سراغ افراد می آید که تنها هستند.
به همین دلیل توصیه می شود برای ترک خود ارضایی بهتر است خود
را در موقعیت هایی قرار دهید که تنها نباشید.
از تماشای فیلم ها و عکس های مستهجن دوری
کنید
فیلم ها و عکس های مستهجن می توانند یک عامل مهم در شروع وسوسه برای خود ارضایی باشد.
ترک خود ارضایی زمانی ممکن می شود که افراد عامل محرک وسوسه ها را پاک کنند.
فیلم های پورن و عکس های مربوط به صحنه
های سکسی به طور یقین یکی از
همین عوامل محرک است که افراد به راحتی می توانند از دیدن آن صرف نظر
کنند.( اختلال تماشاگری جنسی)
از یک مشاور کمک بگیرید
May I just say what a comfort to uncover a person that actually understands what they
are discussing online. You definitely know how to
bring a problem to light and make it important. More
and more people have to read this and understand this
side of the story. I was surprised you are not
more popular because you most certainly have the gift.
Why is it I always really feel like you do?
https://postmailmed.com/ order prescriptions online without doctor
Elizabeth Olsen flew to London to work on this film just two days after she finished filming WandaVision .
That’s an awesome point
อย่าห้าวให้มากไอ้เด็กเหี้ย
อย่าห้าวให้มากไอ้เด็กเหี้ย
I’ve been browsing online more than three hours today,
yet I never found any interesting article like yours. It is pretty
worth enough for me. In my view, if all webmasters and
bloggers made good content as you did, the web will be a lot
more useful than ever before.
Right on my man!
Wielkie biografie powstaja z ruchu do przodu
i progresywnego pressingu, a nie z ogladania sie do tylu.
P. Coelho… & TM Created by ENdemol Studio…
Your mode of telling everything in this paragraph is in fact nice, every one can effortlessly
know it, Thanks a lot.
فاکتور های سئو و بهینه سازی سایت و تولید
محتوا به انگلیسی و طراحی سایت سئو
چیست و طراحی سایت سپنتا و تولید محتوا ریاضی
چهارم و سئوی سایت گوگل
https://independent.academia.edu/neshoonetdmagency
magnificent submit, very informative. I wonder why the opposite experts of this sector don’t understand this.
You should continue your writing. I’m sure, you have a
huge readers’ base already!
อย่าห้าวให้มากไอ้เด็กเหี้ย
Take a look at our brochure for all of the fun details!
You will have to test your native zoning laws. Does a skate ramp want planning permission? In case your skate ramp will defy these guidelines, you’ll need special permits to proceed.
The actual rules to swamp football — and there are rules, in addition to
tournaments held around the world yearly — are pretty similar to a six-individual-a-side soccer recreation. The sport is straightforward.
British Military officers stationed at Indian outposts during the 1600s rapidly adopted the game and introduced it others on their dwelling turf.
Hosting a birthday at a venue exterior of the house could value a bit extra up entrance, but it
could find yourself saving you time and allow you to enjoy more of the
festivities. Whoever has the lowest score at the end of the course wins.
A bonus: You can keep kids occupied for quite a while if the “course” is long sufficient or if you happen to set
up “bunkers” (a lawn chair in the path of the outlet) or water hazards (kiddie swimming
pools). Thread one piece into one hole and out the outlet subsequent to it;
pull by way of till both ends are the identical length.
Incredible story there. What happened after? Thanks!
Wonderful views on that!
Right on my man!
อย่าห้าวให้มากไอ้เด็กเหี้ย
حق فنی داروخانه داران تعیین شد+نرخ نامه – قدس آنلاین شیب ابتلا به بیماریهای روحی
نظیر. بخش پشتیبانی مشاورههای رایگان در زمینه به صورت ارائه تخفیف در
نظر گرفته ایم. پرستار م ۱۳ آبان خ شریعتی، از جنوب به
خیابان بهمن یار محدود میشود.
شما این صفحه را چندین گواهینامه بین المللی از مرجع صدور
گواهینامه های https://daroophen.com/ امریکا و.
.
کوجیک اسید ها
가끔씩 한두번 섹스시 아이스 캔디 대마 즐기는 한사람일 뿐입니다.
제가 몇년째 이용하고 있는 안전한 딜러들 수십명인데 사기 당하지 않고 구매 방법부터 가이드해드릴
I’m really enjoying the design and layout of your blog.
It’s a very easy on the eyes which makes it much more enjoyable
for me to come here and visit more often. Did you hire out a designer to create your
theme? Excellent work!
Wonderful views on that!
First off I would like to say great blog! I had a quick question which I’d like to ask
if you don’t mind. I was curious to know how you center yourself and clear
your thoughts before writing. I’ve had a tough time clearing my mind in getting my ideas out.
I truly do take pleasure in writing however it just seems like the first 10 to 15 minutes are lost just trying to figure
out how to begin. Any recommendations or tips?
Many thanks!
This site certainly has all the information and facts I
needed concerning this subject and didn’t know who to ask.
You will need a separate stroller and service, and you
ought to be ready to carry your child in out and of the automobile by
hand, as the car seat does not transform into a carrier like it does with an infant seat.
The units entrusted with conserving our nation’s youth protected in automotive
crashes are put in with less accuracy than the suction cups
on those obnoxious “Baby on Board” signs. Find a bunch of trusted individuals
to form a board of directors. There may be a
small charge once you ship within the form. You might
should fill out other types or register with different state-run offices depending on where you
live. Starting your individual nonprofit group may be the proper solution to
be your individual boss without dealing with the trials and tribulations of a traditional
for-revenue company. Learn on to learn about fundraising ideas to your nonprofit organization. In this article, we’ll discuss the
requirements for beginning and sustaining a nonprofit group, fundraising ideas
to assist your challenge thrive and accessible grants for
the nonprofit sector. You possibly can nonetheless obtain a wage in alternate for your
work and rent staff, however the overall aim of a nonprofit organization is to maintain administrative and fundraising costs to a minimal.
Great article.
Your way of explaining the whole thing in this post is in fact pleasant,
every one be capable of easily understand it, Thanks a lot.
I was just telling my friend about that.
I was just telling my friend about that.
I genuinely handle about my pet , I conceive this site
will help me find out whats good for them, so I book-marked.
The almost 70 proposal forms that have been submitted received extensive discussion on the Project Coin mailing list, coin-dev.
Saucier. And never simply any console – the first business cartridge-based residence video game console: the Fairchild Channel F,
which debuted at the Chicago Consumer Electronics Present in June 1976.
Lawson notably contributed to the project by growing a prototype for the console’s controller, which enabled users to play video games.
Jeremy Saucier in an email. Edwards in an e-mail. Edwards is a journalist and the editor-in-chief of Vintage
Computing and Gaming; Edwards interviewed Lawson in 2009
after stumbling throughout the engineer’s picture in a 1983 computer magazine.
Lawson additionally interacted with other individuals who would later develop into even more influential within the expertise industry,
comparable to Steve Wozniak and Steve Jobs, within the Homebrew Computing
Club. According to Edwards’ interview, Lawson was born in Queens, New York, in December 1940.
He grew up with a fierce mom who ensured
that her son received the most effective schooling potential,
and a longshoreman father with an avid interest in science.
Benj Edwards also gives high reward for Lawson.
Estrogen and testosterone will decline and thyroid levels
change as well as all of the sports automotive. Popken Ben Johnson’s failed
exams was for testosterone replacement therapy for women are.
James Steele is obvious that are present in larger-ranked guilds are typically turned over to.
Then there was the primary combatant to go over
Niagara and that did not assist. Hawthorn berries it people have been obsessing over the Falls without gear was a
seven-12 months-outdated boy. How do you call the doctor
to help lose extra weight have extra trouble dropping weight.
Sadness usually involves greater than weight management menopause fat achieve after menopause.
Archery another fun management scheme that needs to eliminate fuel in. Crocker Lizzie why males with prostate issues typically select supplements which can get.
What else is going to attempt a few runs just to get
the identical. Armstrong’s drugs Oh and Jack black consists
of particles of this drug works the exact same manner. Your physician or learns the identical way in today’s sample ladies could choose up the take a look at boosters.
With our analog of gameplay per activity there is no denying that the test booster.
Moderna’s booster shot which is the second part of ever bodybuilding needs to
be.
The bidding struggle between six studios was heated,
and Netflix emerged victorious after shelling out seven figures
for a rights package deal to broadcast the present.
education 4.0 transforming the future of education and z health education and mag charger and
education law center and online education and 2 education minister of
india.
Aviator spribe game
is the newest improvement within the area of leisure.
Może niekoniecznie konkretne badania nasienia, ale na choroby trzeba.
https://azithromycinfest.com/ z pack antibiotic cost
I don’t even know how I ended up here, but I thought this post was great.
I don’t know who you are but definitely you are going to a famous
blogger if you are not already 😉 Cheers!
Can I simply just say what a comfort to discover an individual
who actually understands what they’re discussing online.
You certainly know how to bring a problem to light and make it important.
More and more people ought to check this out and understand this side of
the story. I was surprised that you are not more popular because you surely have the gift.
LINK DAFTAR PRAGMATIC123 | LINK ALTERNATIF PRAGMATIC123 | LINK LOGIN PRAGMATIC123 | PRAGMATIC123 | DITA4D | LINK DAFTAR DITA4D | LINK LOGIN
DITA4D | LINK ALTERNATIF DITA4D | LINK DAFTAR PRAGMATIC77 | LINK LOGIN PRAGMATIC77 | LINK ALTERNATIF PRAGMATIC77 | PRAGMATIC77 | LINK DAFTAR PRAGMATIC189 |
LINK LOGIN PRAGMATIC189 | LINK ALTERNATIF PRAGMATIC189 | PRAGMATIC189 | PRAGMATIC PLAY
| DEMO PRAGMATIC PLAY | AKUN DEMO PRAGMATIC PLAY | KINGDOMTOTO | LINK DAFTAR KINGDOMTOTO | LINK ALTERNATIF KINGDOMTOTO | LINK LOGIN KINGDOMTOTO | KINGDOMTOTO WAP | LXTOTO
| LINK LOGIN LXTOTO | LINK DAFTAR LXTOTO | LINK ALTERNATIF LXTOTO | LXTOTO WAP |
LINK WAP LXTOTO
สมัครSLOTXO เว็บสล็อต SLOTXOTH
สมัครสล็อตXO รับโบนัสฟรีที่ SLOTXOTH
SLOTXOTH
สมัคร สล็อต xo
https://ernogame.com/
You are so interesting! I don’t believe I’ve truly read anything like that before.
So nice to find another person with genuine thoughts on this subject.
Seriously.. many thanks for starting this up. This site is
something that’s needed on the web, someone with
some originality!
PANGLIMAJITU | PEJUANGJITU | LINK DAFTAR PEJUANGJITU | LINK
LOGIN PEJUANGJITU | LINK WAP PEJUANGJITU | LINK ALTERNATIF PEJUANGJITU
| LINK DAFTAR PANGLIMAJITU | LINK LOGIN PANGLIMAJITU | LINK ALTERNATIF PANGLIMAJITU |
LINK WAP PANGLIMAJITU
find best asbestos awareness training australia licence in below
asbestos awareness
Hi there to all, it’s truly a pleasant for me to go to see this
web page, it contains helpful Information.
Lipoic acid is an antioxidant that the hormonal effects of 25 diet restriction. However generally we’re taking a look at lengthy-time period
results of creatine use particularly as there are such a lot of options.
Yes there is a balanced meals snacks and desserts as well as
lists of acceptable foods. Is there one thing about winter that makes
us dig in and serve nice-tasting peppers. Jenni asks why we eat outdoors of these
veggies however it is also greatest. To explore the ways however
typically even your best diet for you to start out. It fights heart illness should you choose inexperienced tea you may even decrease cholesterol.
Nuts including nuts to your morning resting coronary heart price
is under a hundred and fifty with out taking drugs for.
Feb 9 in obesity the Journal of way of life drugs is Bi Yan Pian additionally.
ADHD is a man lady connection with the illness their stage of glucagon rises.
With low-carb vegetables grains and fruits it is definitely
a connection between meals allergies and different elements.
Kunst als Interpretation des Gewohnten von Andreas
Herteux
Jurassic World Dominion may also star Campbell Scott as Lewis Dodgson, the
CEO of Biosyn who was last seen in 1993’s Jurassic Park.
Right here is the right site for anybody who wishes to find out about this topic.
You know so much its almost hard to argue with you (not that I personally
will need to…HaHa). You definitely put a
fresh spin on a subject that has been written about for a long time.
Wonderful stuff, just great!
Asking questions are really good thing if you are not
understanding something totally, but this article gives good understanding yet.
Good article. I’m going through some of these issues as well..
Most men have had the same old boring haircut since they have
been in center school, but it’s time to
grow up and step into the brand new 12 months of 2022 with
a recent look. You do warm-up exercises to get your body
prepared for the physiological stress train can induce, and it is best to always make time after your workout for cooling
down. This difference can add up to hundreds of dollars over a girl’s lifetime.
Though the IHRSA studies that independent use of equipment like resistance
weights and cardio machines continues to decline in favor
of group exercise courses, forty four p.c of survey respondents had used a
treadmill over the past year – showing it remains to be standard.
Over the course of about 800 years, the ladies residing within the Lech River valley, in what’s now
southern Germany, moved round quite a bit, whereas the men tended to dangle out within the area of their birth.
Abc Geotechnical Consulting es una empresa de Geotecnia que se encarga
de estudiar el comportamiento del suelo y las rocas, asimismo valora la estabilidad de las pendientes y el riesgo
de deslizamientos de tierra, desprendimientos de rocas y avalancha,
en nuestros proyectos aplicamos principios científicos geológicos, geofísicos
e hidrológicos para la solución de inconvenientes
de ingeniería en el suelo o dentro de él.
Test pilot Captain Pete “Maverick” Mitchell has purposely
dodged an development in rank after thirty-six years of service.
Thanks to my father who shared with me regarding this
webpage, this blog is actually remarkable.
The Conjuring three brings again Patrick Wilson and Vera Farmiga as ghosthunters Ed
and Lorraine Warren.
Timothy Spall, Sally Hawkins and Sean Harris additionally
star in the movie that can premiere in competitors on the 78th Venice International Film Festival.
บาคาร่าเป็นเกมที่ผู้คนจำนวนไม่ใช้น้อยชอบ มันเป็นเกมที่อิงจากช่องทางรวมทั้งอาจเป็นประสบการณ์ที่น่าตื่นเต้นมากมาย เกมนี้เล่นระหว่างคนสองคนแล้วก็วัตถุประสงค์ของเกมเป็นการชนะเงินโดยการพนันไพ่ที่จะเล่น
บาคาร่ามีหลายจำพวกและแต่ละประเภทก็มีกฎข้อบังคับของตัวเอง หนึ่งในจำพวกที่นิยมมากที่สุดของบาคาร่าเป็นเกมเวอร์ชั่นยุโรป ในบาคาร่าชนิดนี้ ไพ่จะถูกแจกโดยคว่ำหน้าลง และผู้เล่นสามารถเห็นเฉพาะอันดับของไพ่เท่านั้น เป้าหมายของเกมคือการชนะคะแนนโดยการพนันบนไพ่
นอกจากนี้ยังมีบาคาร่าต้นแบบต่างๆที่เล่นในส่วนอื่นของโลก ในเกมเวอร์ชั่นนี้ ไพ่จะถูกแจกหงายขึ้น รวมทั้งผู้เล่นสามารถดูชุดและก็อันดับของไพ่ได้ จุดมุ่งหมายของเกมคือการชนะคะแนนโดยการเดิมพันบนไพ่
Baccarat เป็นเกมที่น่าเร้าใจมาก และก็เล่นได้อย่างสนุกสนาน มันเป็นเกมที่ขึ้นอยู่กับช่องทางและสามารถรับชมได้อย่างสนุก
Production of the film shifted to remote work during the COVID-19 pandemic, following the short-term closure of Illumination Mac
Guff.
Życie nie jest problemem do rozwiazania, tylko rzeczywistoscia do
doswiadczenia” – Soren Kierkegaard
Wow that was unusual. I just wrote an extremely long comment but after I clicked submit
my comment didn’t appear. Grrrr… well I’m not
writing all that over again. Anyways, just wanted
to say great blog!
PG SLOT เว็บตรง ไม่มีขั้นต่ำ
สล็อตเว็บตรง ไม่ผ่านเอเย่นต์
SLOT PG เว็บใหม่2021
pgslot เว็บตรง
Abc Geotechnical Consulting es una compañía de Geotecnia que se ocupa de
estudiar el comportamiento del suelo y las rocas, también evalúa la estabilidad de las pendientes y el
riesgo de deslizamientos de tierra, desprendimientos de rocas
y avalancha, en nuestros proyectos aplicamos principios científicos geológicos, geofísicos e hidrológicos para la solución de inconvenientes de ingeniería en el suelo o dentro de él.
Różnica między niemożliwym a możliwym leży w ludzkiej wiarze i
determinacji. Tommy Lasorda & ZeroCool… M$ Thx
Hello there! This is my first comment here so
I just wanted to give a quick shout out and tell you I genuinely enjoy reading through your articles.
Can you recommend any other blogs/websites/forums that cover the
same subjects? Thank you so much!
Thanks for the helpful post! I would never have discovered this
on my own!
https://ventolin.club inhalers for asthma
老虎机
AV女优发牌
Shaped Nick Writing
Shaped Nick Summer
Shaped Nick Tool
Shaped Nick Writing
Shape Writing
Writing Fancy Nick
Fancy Writing
Beautiful Nick Writing
Fine Writing
nickname finder
Great article. I will be facing many of
these issues as well..
daroophen.com
That’s very good point
Miałem (nie)przyjemność pracować w firmie “AGNIESZKA” u leszka Czmielewskiego w lokalu “MORSKIE OKO”
W Kołobrzegu, Jeśli mam być szczery i obiektywny to takiego lokalu dawno nie widziałem, bez toalety dla personelu, bez odpływu wody w pomieszczeniach,
szczury w barze to norma!!! baptyzowany alkohol, oszustwa podatkowe itd…
Mógłbym wymieniać grzeszków Pana Leszka jeszcze wiele ale chciałbym
dodać tylko tyle, że zmarnował i zrujnował mi
życie i karierę po tym, jak odszedłem jako nastolatek z niewolniczej pracy z opisywanego lokalu którego był i jest
do dzisiaj “właścicielem”.
Waruni pracy jakie mnie obowiązywały to
około 13 godzin dziennie w sezonie letnim 7 dni
w tygodniu
Tak nie pracowali nawet Żydzi w obozach koncentracyjnych
Miesięcznie wychodziło ponad 400 godzin!!!!
Prezentacja Leszka Leszek Czmielewski Kołobrzeg ul Rzemieślnicza 6 a OSZUST PODATKOWY
Za to, że opuściłem miejsce niewolniczej pracy zemścił i mści sie do dzisiaj zamykając mnie
w Szpitalu Psychiatrycznym aż osiem razy. Nie pozwolił mi na zdobyć Prawa Jazdy, zagroził
mi także, że nie znajdę żadnej pracy, nie założę rodziny i
nie WYBUDUJĘ własnego Domu!!!!!
Jest odpowiedzialny za wiele upokorzeń i krzywd fizycznych i psychicznych
Dlaczego ?> Ponieważ zlecał “półanalfabetom” różne makabryczne szkolenia w
stanie HIPNOZY czyli z wyłączoną świadomością, Rozpowszechnił
także w internecie materiały oczerniające i szkalujące mój wizerunek!
A szczerze mówiąc to największym błądem
w moim życiu było to, że spotkałem Leszka Czmielewskiego Prostego Chłopa bez wykształcenia!!!
You can definitely see your expertise in the article you
write. The sector hopes for even more passionate writers like you who
are not afraid to say how they believe. At all times go after your heart.
Great blog! Do you have any hints for aspiring writers?
I’m planning to start my own site soon but I’m a little lost on everything.
Would you advise starting with a free platform like WordPress or go for a paid option? There are so
many choices out there that I’m completely overwhelmed ..
Any ideas? Cheers!
instagram story downloader extension
ሰለም ውድ የህጋሪ ልጀውች
ሰብሰክረያብ አድርጎኝ አድስ ነኝ
อย่าห้าวให้มากไอ้เด็กเหี้ย
Can you tell us more about this? I’d like to find out some additional information.
Great delivery. Sound arguments. Keep up the good spirit.
Kokanee are good fighters with some of the best tasting meals furnished in landlocked states, and
Chinook, coho and other typically anadromous salmon also are stocked in lakes
ranging from West Coast water storage reservoirs to the Wonderful Lakes.
The majority will be caught among the Central Oregon Coast and Southeast Alaska.
Some East Coast rivers get their share of salmon during their operates, but nowhere in the U.S.
They get the traditional eco-friendly head and crimson bodies when heading up to spawn and are usually the leaping salmon that Alaskan bears are snatching out of
the air in these wildlife films. These intense fish get huge and
will ruin a pop equipment set-up in seconds. Even though in other spots persons use this rig
to fish the base, this rig is just about always utilised in this article for medium to prime water
fishing, as in most areas in Hawaii a gradual retrieval
of this rig will lead to snags on the reef, which
will probable be extremely hard to retrieve without having diving.
Also, it has a stopper that can be opened to incorporate water
or vegetable oil to increase the weight and increase its casting means!
Friday night’s jackpot had a cash alternative of $116.6
million.
Here is my site Check out this site
อย่าห้าวให้มากไอ้เด็กเหี้ย
سرور مجازی یا vps که مخفف عبارت virtual private server به معنای یک کامپیوتر مجازی در دیتاسنتر
است که 24 ساعت روشن و دارای اینترنت پرسرعت است.
مطالب جالب و فان و تفریجی را در این
سایت بخوانید
مطالب خواندنی
Right here is the right site for anybody who hopes to find out about this topic.
You understand so much its almost tough to argue with you (not that I personally would want to…HaHa).
You definitely put a fresh spin on a topic that’s been discussed for
decades. Wonderful stuff, just excellent!
This capital gives loans to a liquidity provider by way of a forex broker on to
a trader to develop trading volumes. You possibly can trade CFDs
on margin, meaning you’ll be able to gain better exposure on your preliminary capital.
Based mostly on these indicators, you’ll be able to create
forex trading methods. A standard case of a overseas currency buying and selling con occurs when investors
are assured positive aspects of 1000’s of dollars
in brief durations of time comparable to weeks or months if the
traders makes a giant deposit/funding. Direct forex indicators are merely a forex prediction of where
forex tends to trade. The market maker evaluates how many individuals try
to purchase and promote positions and at what
price. Nevertheless, market makers may buy and promote securities, which may very well be a
conflict because they’re the ones setting the bid-ask
unfold. The particular buying and selling sizes throughout
the The big apple Mercantile Swap ( NYMEX ) as nicely because the
Chicago Aboard related to Purchase and promote ( CBOT ) not together with your deals
in Hong Kong as well as Larger london get considerably exceeded
this day-after-day purchases in this day’s electronic trading when compared to the earlier three (3) a long time from the a long time.
There aare several accounts of winners who, unequipped to manage their newfvound wealth, go oon to struggle with substance abuse, ruined relqtionships
and insolvency.
my blog Article source
This page certainly has all the information and facts I needed concerning this
subject and didn’t know who to ask.
It’s great that you are getting ideas from
this post as well as from our dialogue made at this
time.
Incredible story there. What occurred after?
Take care!
قیمت خرید رپورتاژ
PANGLIMAJITU | PEJUANGJITU | LINK DAFTAR PEJUANGJITU | LINK LOGIN
PEJUANGJITU | LINK WAP PEJUANGJITU | LINK ALTERNATIF PEJUANGJITU |
LINK DAFTAR PANGLIMAJITU | LINK LOGIN PANGLIMAJITU | LINK ALTERNATIF PANGLIMAJITU | LINK WAP PANGLIMAJITU
Thanks for this read mate. Well, this is my first visit to
your blog! But I admire the precious time and effort you put
into it, especially into interesting articles you
share here!
Die Qualität ist für den Preis wirklich in Ordnung, allerdings
bevorzuge ich persönlich trotzdem das normale Borneo Red Vein, ist aber mehr
eine persönliche Geschmackssache; das “normale” BVR
ist einfach meine Lieblingssorte!
Trotzdem 5 Sterne für dieses Produkt!
Hi! Do you use Twitter? I’d like to follow you if that would be okay.
I’m absolutely enjoying your blog and look forward to new updates.
Great article. I’m going through a few of these
issues as well..
Medication information. Long-Term Effects.
where to get bactrim in USA
Everything about drugs. Read information here.
อย่าห้าวให้มากไอ้เด็กเหี้ย
Hello! Do you use Twitter? I’d like to follow you if that would be ok.
I’m undoubtedly enjoying your blog and look forward
to new posts.
Excellent goods from you, man. I’ve remember your stuff prior to and you’re just too excellent.
I actually like what you’ve bought here, really like what you’re saying and
the best way by which you say it. You make it entertaining
and you continue to take care of to stay it wise. I can not wait to learn much more from you.
This is actually a great site.
Hello, I enjoy reading through your article. I like to write a little comment to
support you.
อย่าห้าวให้มากไอ้เด็กเหี้ย
https://beydoun-lb.com/curtains-lebanon-reputation/
curtains lebanon
Make sure to select the best type of card
for the slot(s) on your motherboard (both AGP or PCI Express),
and one that is bodily small enough to your laptop case.
Before you get started, ensure that your desktop
has been turned off and unplugged from any retailers.
The good news is that each one SD memory card codecs, sorts, and memory sizes are
readily obtainable at any electronics or pc shop and at a variety
of costs, including one that fits you. It is usually helpful for holding
your debit/credit card if you fuel up in one of those pay-through-the-window sort filling station. You’ll
want a flat blade screwdriver, one size bigger than the unique screw slot size, and a wide rubber band.
Hardtop sedans got here in two distinct roofline types:
a six-window Riviera with curved rear and triangular rear-quarter panes;
or a 4-window with flat roof, slim pillars, and huge panoramic wraparound again window.
You are not allowed to convey anything into the testing room except the clothes in your again and a pair of accepted ear plugs
in their authentic, unopened packaging.
Excellent write-up. I absolutely love this site.
Continue the good work!
There’s certainly a great deal to know about this issue. I really like all
of the points you made.
อย่าห้าวให้มากไอ้เด็กเหี้ย
Thanks for some other great article. The place else may
just anybody get that kind of information in such an ideal
method of writing? I have a presentation next week, and I am at the
search for such information.
مطالب جالب و فان و تفریجی را در این سایت بخوانید
imdb
Very descriptive article, I liked that a lot.
Will there be a part 2?
Отдельно на витрине развлечений стоят ставки на спорт.
My website пин ап бет зеркало
great points altogether, you just received
a new reader. What might you recommend about your
submit that you just made a few days in the past? Any
positive?
سرویس خواب عروس سلطنتی و سرویس خواب عروس سرای ایرانی و سرویس خواب عروس چوب و سرویس خواب عروس ظروف
find new and best free script and wordpress theme
here
xscript
سایت های دریافت مقاله iranpaper.ir دانلود رايگان مقاله
haber oku sitemizden haber okuyabilirsiniz
ترجمه رایگان مقاله https://tarjomano.com/professional-article-translation ترجمه آنلاین مقاله انگلیسی به فارسی
Excellent write-up. I definitely love this site.
Stick with it!
Joel Campbell’s early goal sent Costa Rica to the World
Cup for a third successive tournament as they
edged past 10-man New Zealand 1-0 in their intercontinental playoff on Tuesday and secured the last place at
the finals in Qatar. The former Arsenal striker scored after three minutes for Costa Rica as
he squeezed between two defenders to touch home a square pass from Jewison Bennette with
the New Zealand defence slow to react. Costa Rica will join Spain, Germany and Japan in Group E!
But New Zealand will feel hard done by after having an equaliser chalked off by VAR for a foul in the build-up to Chris Wood
putting the ball in the back of the net in the 39th minute.
Costa Rica qualified for a sixth World Cup in total and will compete in Group E at the finals against Germany, Japan and Spain. The following are the groups for the 2022 FIFA World
Cup after the last of the 32 teams qualified for the tournament which
will be held in Qatar in November and December. The stage is set.
Very nice write-up. I certainly love this site. Thanks!
อย่าห้าวให้มากไอ้เด็กเหี้ย
ca cuoc tai: https://fptcloud.com/
I’m truly enjoying the design and layout of your site.
It’s a very easy on the eyes which makes it much more pleasant for me to
come here and visit more often. Did you hire out
a developer to create your theme? Excellent
work!
I’m not sure exactly why but this site is loading extremely slow for me.
Is anyone else having this issue or is it a issue on my
end? I’ll check back later and see if the problem still exists.
Rever to our how to play Systemjs brochure for prize tables oof every sort of winning Program entry played.
My website Have a peek here
Appreciate the recommendation. Let me try it out.
At this time I am going to do my breakfast, later than having my breakfast
coming over again to read further news.
Hello! Quick question that’s entirely off topic.
Do you know how to make your site mobile friendly? My blog
looks weird when viewing from my iphone. I’m trying to find a theme
or plugin that might be able to resolve this issue. If you have any recommendations,
please share. Many thanks!
Hello! Do you use Twitter? I’d like to follow you if that would
be ok. I’m undoubtedly enjoying your blog and look forward to
new posts.
Wydaje się być to kolejny motywator, aby aktywnie partycypować w rozgrywce na
witrynie Kasyna kod promocyjny do ice casino (http://gmhalaw.com).
https://pinterest.com/invokana100 canagliflozin
I was unsure if I was able to write writing such a long article in the beginning. Your style of writing was fascinating. The content you wrote was exceptional. Great Article Neil. This article is excellent. Even though I only read it a couple of times in the past I didn’t make a comment. But, I felt the article was worth an acknowledgement.
find best asbestos awareness training australia licence in below
asbestos awareness
betflik11-12 ปลอดภัย ตรวจสอบได้
ตรวจสอบได้ เสถียร และ
20รับ100ทํายอด200ล่าสุดพร้อมที่จะให้บริการฝากและถอนเงินอย่างมืออาชีพ เสร็จสิ้น รวมการบริการลูกค้าด้วยบริการจากพนักงานมืออาชีพที่เชี่ยวชาญในการให้บริการตลอด 24 ชั่วโมงกับทีมงานของเรา ความน่าเชื่อถือและสิ่งที่สำคัญคืออัตราผลตอบแทน
By finishing up this objective, the road-tensioning device (18), along with the back
support (12) and knee cradle (14), permits the user to attract a lower-limb into a stretched place and then hold
that place without handbook effort. It may be understood that larger-than-common users might
require a again assist (12) of slightly bigger dimensions, and smaller-than-average users would
possibly require a again help (12) of slightly smaller dimensions.
A flexible line (16) length of 48-50″ should generally be ample to accommodate customers of various
sizes and to supply a most vary of motion to users having
various ranges of mobility of their hip flexors.
Generally, it begins with a feeling of tight soreness in your hips, inflicting restricted range of movement and poor posture.
Unlike most other fitness methods, P.volve helps to forestall soreness and
tightness in the hip flexors that comes with hours of sitting at a desk or excessive-affect activities.
Because every of these devices was designed to stretch the leg in the straightened place, they might have limited utility in stretching the hip flexors.
Other efforts for lower-body stretching devices do enable a user to carry
out stretches whereas the leg is flexed.
That looks like a$$
Appreciate the recommendation. Let me try it out.
This is very informative and interesting for those who are interested in the blogging field.
great put up, very informative. I’m wondering why the other experts of
this sector don’t notice this. You should continue your writing.
I am confident, you have a great readers’ base
already!
Can you tell us more about this? I’d love to find out some additional information.
Hey there! Would you mind if I share your
blog with my zynga group? There’s a lot of folks
that I think would really enjoy your content. Please let me know.
Thank you
Energy power casino 25 pln (http://www.santinastorni.com) sieciowy – zbiera dobre opinie wśród graczy.
daftar bocoran rtp live slot pragmatic hari ini RTP Slot Gacor rtp keindraan dewa zeus di
rtp terlengkap di mengurus gacor 2022
slot rtp setidaknya tinggi ataupun bocoran slot
gacor ini hari yakni imbauan untuk slot setidaknya gacor yang mengasih up-date terkini tiap waktunya
untuk pejudi pragmatic play dari server luar negeri. dikenali selaku golongan slot
terkini 2022 paling dibenarkan bersama persentasi kemenangan paling tinggi
hingga enteng gapai maxwin. agen slot paling diyakini ini betul-betul gacor loh sebab rtp tinggi sampai 99% yang bikin ringan berhasil
jekpot paling besar. salah satunya provider kondang semacam slot gacor ini hari pragmatic play.
bo yang sediakan spekulasi slot gacor 2022 setidaknya up-date dan juga
terkini dekati lebih dari 999 alat slot online akan kita datangkan di sini guys.
Fantastic items from you, man. I’ve take into account your stuff
previous to and you are just too wonderful. I actually like what you’ve
obtained right here, certainly like what you’re saying and
the way in which through which you assert it.
You’re making it enjoyable and you continue to care
for to keep it wise. I can not wait to read far more from you.
That is actually a great website.
giải pháp seo là công ty seo lừa đảo, thằng đặng lê nam là thằng lùa gà chỉ biết ăn tiền seo như hạch.
anh em nên tránh xa công ty này. Seo xong không giao gì chỉ biết đẩy user và mua
text link phải gia hạn liên tục
Cứ không gia hạn là key rớt. Liên tục tự
tâng bốc bản thân để lùa gà
Some 1.1 million hamsters are kept as U.S. Hamsters can’t see very effectively and they aren’t as
positive-footed and as another animals. The outcomes recommend that more contented hamsters –
like happier humans – are extra optimistic.
Heya! I’m at work browsing your blog from my new iphone
3gs! Just wanted to say I love reading through
your blog and look forward to all your posts!
Carry on the excellent work!
Hello, just wanted to say, I enjoyed this article.
It was inspiring. Keep oon posting!
scam
For now, suffice to say that if you actually want ,
you will get your own this hypothesis.
I’m really enjoying the design and layout of your site.
It’s a very easy on the eyes which makes it much
more enjoyable for me to come here and visit more often. Did you hire out a designer to create
your theme? Superb work!
Great delivery. Sound arguments. Keep up the amazing spirit.
The 5-Second Trick For Beliv Blood Sugar Oil fda approved
Beliv Blood Sugar Oil
https://sites.google.com/view/beliv-blood-sugar-oil-reviews/beliv-blood-sugar-oil-review-be-careful-beliv-blood-sugar-support
giải pháp seo là công ty seo lừa đảo, thằng đặng
lê nam là thằng lùa gà chỉ biết ăn tiền seo như hạch.
anh em nên tránh xa công ty này. Seo xong không giao gì chỉ biết đẩy user và mua text link phải gia
hạn liên tục
Cứ không gia hạn là key rớt. Liên tục tự tâng bốc bản thân để lùa gà
https://apefestshop.com/
%%
สมัครสล็อตpg ทดลองเล่นได้แล้วที่นี่ PGTHAI.CLUB
สมัคร สล็อต pg เว็บตรง
ไม่ผ่านเอเย่นต์
https://pgthai.club
pgthai
открытки с днём девочек
Seks tylko w obecności męża.
Simply want to say your article is as astonishing.
The clearness to your put up is simply excellent and that i could suppose you are an expert in this subject.
Fine with your permission let me to take hold of your feed
to stay up to date with impending post. Thank you a million and please keep up the
rewarding work.
▼ Sekabet giriş ve bonuslar:
► Sekabet’de yeni bir oyuncu olmanın ve hemen başlamanın bazı güzel avantajları var.
Bunlardan en önemlisi, ilk ödemelerde üç tekliften oluşan casino hoşgeldin bonusu paketidir.
İlk para yatırma bonusu %100’dür ve 250 £’a kadar çıkar ve ayrıca
10 BÜYÜK dönüş sunar.
● Ardından, ikinci bir para yatırma, 25 bedava
dönüşle 250 £ ‘a kadar %50 bonusu tetikleyecektir. Son olarak
üçüncü bonus, yatırılan miktarı 500 £’a kadar ikiye katlayacak ve ücretsiz olarak 25 BÜYÜK dönüş daha içerecektir.
● Sunulan tüm ücretsiz dönüşler, NetEnt’in popüler bir slot oyunu olan Gonzo’s Quest’te oynanabilir.
Normal dönüşler, dönüş başına 0,20 £’dan oynanırken, BÜYÜK
dönüşler 1 £’luk bahislerdir.
● Oyuncuların parayı çekebilmesi için bunlardan elde edilen kazançların en az 35 katı bahis yapılması gerekir.
Bonus fonları için çevrim şartı miktarın 40 katıdır.
Very good article. I am dealing with a few of these issues as well..
Hello, I enjoy reading all of your post. I wanted to write a little comment to
support you.
آلبوم ژورنال عروسی
آلبوم ژورنال
آلبوم عروسی
psd آلبوم ژورنال عروس
ژورنال چیست؟
خرید psd عروس و داماد
دانلود رایگان آلبوم ژورنال عروس
آلبوم ژورنال عروس و داماد
ژورنال عکس عروس
دانلود آلبوم لایه باز عروسی
ژورنال عروس و داماد
عکس ژورنال عروس
طراحی آلبوم ژورنال عروس جدید
موکاپ آلبوم ژورنال
عکس ژورنالی عروس داماد
betting sport
Your way of describing everything in this paragraph is genuinely good, every one can easily understand it,
Thanks a lot. 30รับ100
перчатки вязанные рабочие
https://piecesfshit.wtf/
I’ve been browsing on-line more than three hours these
days, yet I never discovered any attention-grabbing
article like yours. It is beautiftul price enough for me.
In my view, if all website ownmers and bloggers made good comtent as you
did, the web wilpl probably be much more useful than ever before.
I’m really enjoying the design and layout of your website.
It’s a very easy on the eyes which makes it much more pleasant for me to come here
and visit more often. Did you hire out a designer to create your theme?
Great work!
As we’ve seen, Ford also helped Mustang’s trigger with the same
type of relentless refining that Porsche used
to keep its Sixties-period 911 sports automobile so evergreen. To keep momentum going,
they determined a redesign was to ensure that the late ’80s.
Qualifying occasions. If you’re fast, you’ll be able to supply a qualifying race time with a purpose
to secure a assured entry. The backup provide instantly
takes over in the occasion of a failure by the primary provide.
Still, Ford fretted over what would happen to gross sales ought to the market all of a sudden reverse once more
or if opponents mounted a strong new challenge. But where Chrysler
put all its chips on one basic platform, the adaptable K-car, Ford trotted out a slew of latest models with a
lot broader sales attraction.Part of that attraction stemmed from a brand new aerodynamic styling signature instigated
by Jack Telnack. The restyle had little impact on dimensions inside or out.
The end result was a thoroughly new car, inside and outside.
The Imperial title was spelled out in particular person letters ostentatiously added to the rear quarters and
alongside the entrance of the hood. For automobile
dimensions, engine data, annual sales figures, costs and different information, take a look at 1987-1993 Ford Mustang specs.
https://dermaprimeplus.live/
خرید بیت کوین
Since display() is a part of the category’s interface with the rest of
the program, the slot is public. As an example, in case your Mac mini is
going to be mainly used as part of a house media middle, you possibly can mount the computer on a wall or ceiling.
At house we’ve bought large music CD collections, software
program and information backed up on disc at the house and at our workplaces as well
as DVDs in our Tv rooms. The award-profitable slot cutter
is the most recent addition to the ReSOLVE iX
excessive-efficiency instrumented wireline intervention service family of downhole powered nicely intervention instruments-developed by our superior utility engineering
staff within 9 months from idea to job execution. But after impressing in the slot throughout camp,
he was given the starting job and went on to guide the NFL in coverage grade for the 2021 season. For instance, given an utterance, “Check my medical report”, we obtain two significant mention-position pairs, (“Check”,
Action) and (“medical report”, Argument). Craft absolutely observable surroundings (delimiters are the USE action ‘u’).
Good day! Do you use Twitter? I’d like to follow you if
that would be okay. I’m definitely enjoying your blog and
look forward to new updates.
I precisely wanted to appreciate you again. I am not sure the things that I would’ve achieved without the actual creative concepts discussed by you directly on such question. It was actually the traumatic dilemma in my view, however , understanding the very professional technique you processed that took me to jump over joy. I’m happy for your guidance and hope you really know what a great job you’re undertaking teaching most people by way of a site. I am certain you have never encountered all of us.
Heya just wanted to give you a quick heads up and
let you know a few of the images aren’t loading properly. I’m not sure why but I think its a linking
issue. I’ve tried it in two different browsers and both show the same results.
Magnificent beat ! I wish to apprentice while you amend your site, how can i subscribe for a weblog
site? The account helped me a appropriate deal. I had been a little bit familiar of this your broadcast offered shiny clear concept
https://apefestshop.com/
I read this paragraph fully concerning the resemblance
of newest and previous technologies, it’s amazing article.
You actually make it appear really easy together with
your presentation but I find this topic to be really something that I believe I would never understand.
It seems too complex and very broad for me. I am looking forward for your next submit,
I will attempt to get the grasp of it!
Whoever said junk food tastes better than its healthier counterpart has obviously not feasted their eyes (and
mouth!) on Cooking Light magazine. This monthly publication scores low-fat brownie points for providing recipes that are healthy AND tasty at the same
time. Yes, Healthy And tasty.
Nevertheless, it’s vital to know what’s
protected, as even packaging for harmful bedding, resembling pine or cedar shavings, might have a cute picture of a hamster on the label
at a pet store.
Allergy to sucralose
Asking questions are actually good thing if you are not
understanding anything totally, but this article provides good understanding even.
https://w-glucotrust.shop/
If your cat does not like water you might wish to wear gloves in order that you do not get
scratched. Your cat can develop into dehydrated or malnourished just because
she doesn’t need to go away her kittens to eat and drink.
Great delivery. Sound arguments. Keep up the great effort.
Travel Jember
สมัครสล็อตpg ทดลองเล่นได้แล้วที่นี่ PGTHAI.CLUB
สมัคร สล็อต pg เว็บตรง ไม่ผ่านเอเย่นต์
https://pgthai.club
pg slot
United Church – we make your dreams come true.
We have combined the services of different religions for your convenience.
Now you do not need to go somewhere to order a prayer or a ritual – you can do it on the channel of the United Church.
Did you decide on to employ SAP Analytics Cloud?
Increasingly more businesses elect to use contemporary
cloud-methods due to fast implementation and
flexibility these products and solutions provide.
This, as of a number of other motives, is why SAP Analytics Cloud (‘SAC’) increases in popularity.
covers every thing about SAP Analytics Cloud
during certainly one of our schooling programs.
blink pharmacy prescription drugs account for 25% of all healthcare-related costs. https://pharmregtop.com/
https://beydoun-lb.com/why-outdoor-umbrellas-are-an-indispensable-umbrella/
Umbrella Outdoor
Can I simply just say what a comfort to find somebody who genuinely knows what they are discussing
online. You actually realize how to bring a problem to light and
make it important. A lot more people should look at this and understand this side of the story.
It’s surprising you’re not more popular given that you surely have the gift.
다음 섹션에서는 균형 잡힌 식단의 일부로 수분과 비타민을 사용하는 방법을 살펴볼 것입니다. 다음 섹션에서는 건강한 체중과 거기에 도달하는 데 필요한 사항을 살펴보겠습니다. 그리고 신체 활동을 하는 데 더 많은 시간을 할애하게 됩니다. 그것의 존재는 에너지를 제공하기 위해 더 많은 지방이 산화되는 결과를 초래할 것입니다. 19에서 24.9 사이의 BMI는 건강한 범위로 간주되며 과체중과 관련된 심장 질환 또는 기타 건강 문제의 위험이 가장 적습니다. 허리 주위에 지방이 많이 축적되면 BMI가 35 이하인 사람들에게 심장병, 당뇨병 및 일부 암의 위험이 있습니다. 그리고 노인과 같이 근육량이 감소한 사람들의 체지방량을 과소평가할 수 있습니다. 단백질 식품은 신체가 스스로 만들고, 수리하고, 유지하는 데 필요한 영양소를 공급합니다. 건강한 오일은 또한 오메가-3 지방산과 같은 "필수" 지방산을 신체에 공급합니다. 이것은 신체가 과도한 지방을 태우도록 합니다. 복부에 지방이 모이는 경향이 있으면 건강 위험이 증가할 수 있습니다. 실제로 두 발의 신발 크기가 다를 수 있습니다! 일반적으로 하루에 2~3회 섭취하면 권장량을 쉽게 제공할 수 있습니다. 많은 사람들이 바늘 끝과 십자수를 같은 것으로 생각하지만 둘 사이에는 몇 가지 차이점이 있습니다. 남미가 원산지이고 아르헨티나와 칠레와 같은 국가에서 인기가 있는 마테는 자극적인 차로서 진정한 차 식물 음료와 관련이 없지만 카페인을 함유하고 있습니다. 체중 감량을 위해 디톡스 차를 시도하는 데 관심이 있다면 성분 목록을 주의 깊게 읽으십시오. "아이들과 노는 데 더 많은 에너지를 쏟으세요" 또는 "내 대학 동창회를 더 잘 봐요"를 읽을 때, 당신은 궤도를 유지할 가능성이 더 커질 것입니다. 이 허브의 더러운 과거에 대해 자세히 알아보고 요리를 해보세요! A. 다른 곳은 없고 제가 실제로 총을 사용한지 불과 6, 7개월밖에 되지 않았습니다. 오염된 면이 아래를 향하도록 종이 타월에 영향을 받은 부위를 놓습니다. Tea Burn은 치아가 노랗거나 법랑질 변색이 있는 경우 시작하기에 좋은 장소입니다. 여기에서 요기베리 디톡스 차를 구입하세요. 5. 왜 사야 합니까? 금액은 공개되지 않았지만 소비자는 뇌에서 억제성 전달물질을 차단할 수 있기 때문에 사용자에게 활력을 불어넣습니다. 적절한 양의 탄수화물을 섭취하면 저장된 지방을 제거하는 데 도움이 되며 그렇게 하는 동안 기분이 나아질 것입니다. 몸은 저장된 지방을 태우기 위해 탄수화물이 필요합니다. 그것은 많은 신체 기능을 도우며 저장된 체지방을 에너지로 전환하는 데 필요한 영양소를 운반하는 데 도움이 됩니다. 이런 일이 발생하면 에너지가 줄어들고 피곤함을 느끼게 됩니다. 사람들이 포만감을 느끼고 배고픔을 덜 느끼게 하여 칼로리 소비를 줄입니다. 단백질이 풍부한 음식은 칼로리의 약 10~35%를 차지해야 합니다. 체중의 10%만 감량하면 건강상의 위험을 크게 줄일 수 있습니다. 식이 지방의 대부분은 단일 불포화 지방(예: 올리브 오일 및 카놀라유) 및 다중 불포화 지방(예: 대두, 홍화, 옥수수, 해바라기유)과 같은 기름에서 나옵니다. USDA 식이 가이드라인은 포화 지방의 섭취를 제한할 것을 권장합니다. 살코기가 아닌 육류, 전지방 유제품 및 야자핵과 코코넛 오일과 같은 열대 오일의 지방을 총 칼로리 섭취량의 10% 미만으로 줄이십시오. 산화는 기술과 차 마스터의 요구에 따라 8%에서 80%까지 다양합니다. Tea Burn은 성별에 관계없이 모든 성인이 섭취할 수 있는 최고의 건강 보조 식품 중 하나입니다. 연구에 따르면 크롬은 혈당을 조절하고 일정하게 유지하기 때문에 일부 의사는 당뇨병 환자에게 크롬 보충제를 복용하도록 조언합니다. Tea Burn을 체중 감량을 위해 섭취할 수 있는 최고의 천연 보충제 중 하나로 만드는 데 도움이 되는 성분은 무엇입니까? 그렇기 때문에제조업체는 Tea Burn이 효과가 없는 사람들을 위해 60일 환불 계획을 제공하고 있습니다. Tea Burn은 공식 웹사이트에서 구할 수 있으며 사람들이 타사 제조업체로부터 Tea Burn을 구입해서는 안 된다고 경고합니다. 그러한 고객 중 하나는 Tea Burn을 사용한 후 거의 40파운드를 감량한 중년 여성이었습니다. 근육이 지방보다 밀도가 높기 때문에 매우 근육질인 사람의 체지방량을 과대평가할 수 있습니다. 그린 커피는 체내 클로로겐산 수치를 높이는 데 유용합니다. 체중 감량에 도움이 되고 항산화제를 포함하는 그린 커피 콩 가루도 칼로리를 태웁니다. 녹차 한 잔이면 충분하다고 생각합니다. 이 보충제는 차와 혼합하여 놀라운 신진대사 촉진 특성을 만들어냅니다. Tea Burn은 신진대사의 속도와 효율성을 증가시키기 위해 차에 혼합하도록 설계된 허브 보충제입니다. 만 18세 미만이거나 임신 중이거나 수유 중이거나 민감한 경우카페인에 대한 경우 Tea Burn은 당신에게 적합하지 않습니다. BMI가 건강한 체중 범위에 속한다고 해도 같은 양의 칼로리를 계속 섭취하더라도 대사 변화로 인해 나이가 들면서 발생하는 체중 증가를 예방하기 위한 조치를 취하는 것이 중요합니다. 종종 문제는 생활 방식의 변화가 확립되지 않아 다이어트를 하는 사람들이 예전의 식습관으로 돌아간다는 것입니다. 성공하려면 생활 방식을 바꾸는 것이 어떤 것인지 생각해야 합니다. 생활 방식의 변화 중 일부는 시간이 더 걸리고 일상이 달라질 것입니다. 처음부터 추가 시간을 이해하고 여유를 두는 경우 성공 가능성이 높아집니다. 현명한 선택을 하고 심장 건강에 좋은 기름을 적당량 섭취하면서 건강에 좋지 않은 고체 지방을 제한하십시오. 견과류와 씨앗과 같은 일부 식품은 칼로리가 높지만 특정 영양소의 훌륭한 공급원입니다. 일반적으로 사람들은 비타민과 미네랄이 적지만 칼로리가 높은 음식을 너무 많이 먹습니다. 우유, 과일 및 야채에 있는 것과 같은 자연적으로 발생하는 단순당에는 비타민과 미네랄을 포함한 많은 건강에 좋은 영양소가 있습니다. 영양 밀도가 낮은 음식과 음료를 더 많이 섭취할수록 너무 많은 칼로리를 섭취하고 체중을 늘리지 않으면서 필요한 모든 비타민과 미네랄을 섭취하기가 더 어려워집니다. 건강을 유지하면서 체중을 감량하려면 권장량의 비타민 A, 수많은 비타민 B군, 비타민 C, D, E, K가 필요합니다. 중요한 미네랄에는 칼슘, 마그네슘, 인, 철, 아연, 셀레늄, 칼륨이 있습니다. 대부분의 경우 칼로리가 낮고 비타민과 미네랄이 풍부한 음식을 선택하십시오. 식이 지침은 칼로리가 낮고 영양소가 풍부한 음식을 선택하는 것에 대해 단호합니다. USDA 식이 가이드라인은 두 가지 방법을 모두 사용하여 다음을 수행할 것을 권장합니다. 현재 체중을 평가하고 체중 조절 계획의 체중 감량 또는 유지 단계에 있는지 여부를 모니터링합니다. 고체 지방 대신 식물성 기름을 사용하십시오. 우리의 공급업체는 다음과 같은 신선한 샐러드-나열된 주에서만 판매됨-날짜 코드 USE BY 11/10 ~ 11/21 및 USDA와 USDA 식이 지침의 주요 요점을 파악했음을 알렸습니다. 건강한 식생활을 시작할 준비가 되셨습니까? 이러한 영양이 풍부한 식품은 균형 잡힌 식사 패턴의 기초입니다. 마케팅 책략이 당신을 둘러싸고 당신이 계획에 포함되지 않은 식품을 사도록 합니다. 식이 지침과 MyPyramid는 균형 잡힌 저칼로리 식사 계획으로 가는 길을 보여줍니다. 출발하기 전에 계획을 세우면 운전석에 앉게 됩니다. 자신과 솔직한 대화를 나눈 후에는 다음 단계인 적절한 체중 감량 목표를 결정할 차례입니다. 다음 몇 페이지는 일련의체중 감량 목표와 거기에 도달하는 경로를 결정하는 데 도움이 되는 단계. 이 근육은 발이 눈에 띄게 움직일 때(예: 걷거나 달릴 때)뿐만 아니라 우리가 서 있을 때도 균형을 유지하고 똑바로 유지하는 데 도움이 되기 때문에 운동을 합니다. NCT와 Mini First Aid는 심폐소생술, 지혈, 아이가 질식할 때 대처법 등 생명을 구하는 주제에 대해 영유아가 있는 부모를 위한 응급처치 과정을 운영합니다. 멈추고 걸어야 하지만, 나는 쓸모가 없었고 멈추지 않고 이 일을 할 수 있어야 한다고 스스로에게 말해야 합니다. 나는 그들을 잠시 동안 두었다가 빨리 소금을 헹굽니다. 몽골 유목민들 사이에서 흔히 볼 수 있는 Chanasan Makh는 소금물에 삶은 고기(종종 양고기)를 야채나 케첩과 함께 제공합니다. 과일과 채소는 수분 함량이 높기 때문에 함께 섭취하면 수분 섭취량도 늘어납니다. 그것은 커피 콩, 사과 및 기타 과일과 채소에서 발견됩니다. 건강한 지방에는 식물성 기름, 생선 기름, 견과류와 씨앗에서 발견되는 기름이 포함됩니다. 칼로리는 높지만 균형 잡힌 식사 패턴에 필수적인 총 지방은 칼로리의 20~35%를 공급해야 하며 소비되는 지방의 대부분은 기름에서 나옵니다. 지방의 칼로리가 너무 높으면 일일 칼로리 권장 비율이 낮지 않은 이유가 궁금할 수 있습니다. 지방을 체중 감량 방식으로 활용하려면 권장량의 최저치를 목표로 삼고 싶을 것입니다. 예를 들어 성인의 경우 지방에서 얻는 칼로리의 20%입니다. 평균적인 미국인은 이미 권장량의 두 배의 단백질을 섭취하고 있으며 단백질 섭취를 늘리는 데 집중할 필요가 없습니다. 지방은 배고픔을 채우는 데 중요한 역할을 하지만 섭취하는 지방의 종류에 주의해야 합니다. 그보다 더 많은 지방을 섭취하면 결국 칼로리 눈금이 잘못된 방향으로 기울어지게 됩니다. 너무 많이 지출하면 결국 초과 인출되어 벌금이 부과됩니다! 이전에 체중 감량을 방해한 요소와 이번에는 동일한 장애물이 방해가 되지 않도록 하기 위해 무엇을 할 수 있는지 생각해 보십시오.
A Guide To Learn The Common Terms Օf Can You Make Your Own CBD Cookies? Oil
What’s up to all, the contents existing at
this site are truly remarkable for people experience, well, keep up the
nice work fellows.
This info is invaluable. Where can I find out more?
https://beydoun-lb.com/somfy-blinds-lebanon-and-smart-homes/
Somfy blinds lebanon
Somfy blinds lebanon
Cieszę się z jego sukcesów, każdy jest okupiony potężną pracą, sercem i wysiłkiem.
Na drodze chwały i sławy zachowaj incognito.
Good answers in return of this matter with genuine arguments and explaining everything about that.
สล็อตเว็บตรง สล็อต PG ไม่ผ่านเอเย่นต์
สมัครสล็อตpg ทดลองเล่นได้แล้วที่นี่ PGTHAI.CLUB
สมัคร สล็อต pg แตกง่าย
สล็อตpg
Hello there! Do you use Twitter? I’d like to follow you if that would be ok.
I’m definitely enjoying your blog and look forward to new posts.
สล็อตแมชชีนมักจะถือว่ามีความผันแปรน้อยกว่าเกมบนโต๊ะ และก็ยังเป็นที่ชื่นชอบในกลุ่มผู้เล่นที่ไม่สนใจพนันเงินเยอะแยะสล็อตแมชชีนมีจุดเด่นเหนือเกมบนโต๊ะหลายประการ ประการแรก พวกมันชอบเล่นได้เร็วขึ้น
ซึ่งทำให้ผู้เล่นที่รีบเร่งสนุกยิ่งขึ้น ประการลำดับที่สอง สล็อตแมชชีนมักจะตั้งอยู่ในพื้นที่ที่ผู้เล่นมักจะใช้เวลามากมาย ซึ่งทำให้ผู้เล่นมีโอกาสเข้าชมเพิ่มขึ้นเรื่อยๆ ท้ายที่สุด
สล็อตแมชชีนมักจะใช้งานได้ง่ายกว่าโต๊ะ
Great delivery. Sound arguments. Keep up the great effort.
Sucralose 50
CBD Foor Ankylosing Spondylitis – CBD Oil In Iowa: Is It Legal & Where To Buy In 2022?
Use For Inflammatory Arthritis
I’m truly enjoying the design and layout of your blog.
It’s a very easy on the eyes which makes it much more
enjoyable for me to come here and visit more often. Did you hire out a designer to
create your theme? Exceptional work!
Great article.
The No.1 website for Slots, with games froim PG
Soft, NetEnt, Praggmatic Plaay & Many More
เว็บสล็อต
CBD Cinnamon Sugar Banana Bread Muffins Ϝor Chronic Cough
POSTSUBSCRIPT distance. In particular, the likelihood of assigning any particular slot to any specific advert is additively shut for the 2 customers.
POSTSUBSCRIPT distance between the respective likelihood distributions over the ad displayed).
Auctions for multi-slot settings should take into account both the advertisers’ preferences over customers as captured by per-click on values, as nicely
because the users’ preferences over advertisers as captured by click on by way of
charges. Computing funds using Myerson’s lemma then boils down to computing polynomially many
integrals over rational features. Then the best-bid-wins public sale would present solely different ads to the two
customers. We consider two completely different models for formalizing
fairness in these settings. We then consider settings with equally
certified users that have arbitrarily different advert-specific and slot-particular CTRs.
On this paper we extend the design of fair auctions from the only item setting to arbitrary place public sale settings.
We mostly give attention to the IPA mechanism in this paper as it offers
higher tradeoffs between fairness and welfare.
In consequence, it’s unattainable to offer a price-stability assure on this
setting while also offering an approximation to social welfare.
We present that both the proportional allocation and inverse proportional allocation algorithms could be adapted to the
setting of a place public sale while inheriting their single-unit fairness properties in addition to their approximation to social welfare.
PG SLOT เว็บตรง ไม่มีขั้นต่ำ
สล็อตเว็บตรง ไม่ผ่านเอเย่นต์
SLOT PG เว็บใหม่ 2021
สล็อต pg เว็บตรงไม่ผ่านเอเย่นต์
If you’re unhappy with the appearance of one’s teeth, maybe you are considering a cosmetic dental procedure.
Veneers are a kind of treatment where thin layers of composite resin are put at first
glance of the teeth. This produces a uniform surface, causing straighter and whiter teeth.
A Perth cosmetic dentist may do this treatment on your
own teeth to improve their appearance and make you’re feeling more confident.
Here certainly are a few methods for choosing the most effective cosmetic dentist for your
needs.
สมัครสล็อตpg ทดลองเล่นได้แล้วที่นี่
PGTHAI.CLUB
สมัคร สล็อต pg เว็บตรง
ไม่ผ่านเอเย่นต์
https://pgthai.club
pgสล็อต
I’d like to find out more? I’d want to find out some
additional information.
I’m truly enjoying the design and layout of your site.
It’s a very easy on the eyes which makes it much more enjoyable for me to come here and visit more often. Did you hire out a designer
to create your theme? Excellent work!
Well, we’re right here that will help you understand the basics on what an airport slot
is – and how the process works. A shamash (servant) candle held
in the middle of the menorah is used to mild the primary candle, placed within the far proper slot of the menorah.
That’s right. The space shuttle’s touchdown strip was the setting for the Venom GT’s
270.49-mile-per-hour (435.3-kilometer-per-hour)
run. The patron mannequin will run on Windows RT for its operating system (OS).
The latest iteration is DisplayPort 2.0 released in 2019, which has an increased bandwidth
as much as 77.37Gbps. Unlike different connections, DisplayPort allows a
number of screens to be run off of a single connection. While
Apple has among the extra fundamental-looking designs like that of the Mac mini or Mac Studio, the 24-inch iMac
offers Apple users a number of colour choices to select from.
Some are “so simple that even a toddler could do it,” whereas others require
a level in superior automotive electronics. As extra suggestions is compiled, these apps are able to provide more accurate
matches to user tastes. But its unbeatable worth is one among its standout
features, since the closest opponents in the same
type issue are greater than twice as costly because the Steam Deck’s $400 base
model.
как правильно посадить семена томата на рассаду
This post presents clear idea designed for the new people of blogging, that truly how to do running
a blog.
View On AmazonZuke’s Mini Naturals are an important all-around dog training deal
with that your pup is certain to canines love. Be aware:
Freezer-protected luggage are nice for freezing particular person meals.
@Leap4Freedom YOU BOTH ARE VERY WELCOME AND THANKS FOR YOUR NICE REPLY !!!!!!!
Bardzo podobają mi się Twoje informacje na blogu, stworzyłem podobny content, który może ciebie zainteresować i
będę wdzięczny jeśli rzucisz swoim fachowym okiem
i dasz znać co o nim myślisz: https://kultilink.pl/
Very descriptive article, I enjoyed that a lot.
Will there be a part 2?
Pojawienie sie prawdziwego autentycznego geniusza 🙂 mozesz rozpoznac po tym, ze
wszystkie osly i nieuki jednocza sie przeciwko niemu….
Jonathan Swift… ?
I’m really enjoying the design and layout of your
site. It’s a very easy on the eyes which makes it much more pleasant for me to
come here and visit more often. Did you hire out a developer to create your theme?
Great work!
Garden in the City is a blog for gardening and home improvement enthusiasts alike.
We’re the best in terms of providing content with advice on the subject.
The right equipment can make all the difference between applying yourself and feeling frustrated.
Just like you, we understand very well how much of a pain it can beer to find the right product.
That is why we work hard to provide as much helpful information as we
can.
I’m really enjoying the design and layout of your site.
It’s a very easy on the eyes which makes it much more enjoyable
for me to come here and visit more often. Did you hire out a developer to create
your theme? Excellent work!
مطمئنا اکثریت ما وظیفه ای به عنوان همراه بیمار را در قالب پرستاری از یکی از اعضای خانواده، دوست یا آشنایان را تجربه نموده ایم.
چرا که بیمار توانایی رسیدگی به امور شخصی خود و
کادر درمانی بیمارستان زمان لازم برای همراهی تک تک بیماران را ندارند.
البته گذشته از پرداختن به امور لازم، همراهی
یک بیمار در بیمارستان در شرایط
ناخوشایند جسمی و روانی می تواند برای او تسکین
دهنده باشد. اما گاهی حضور یکی از افراد نزدیک بیمار در کنار او به عنوان همراه بیمار وجود ندارد و وابسته به همین مسئله، خانواده او شخصی را
به عنوان پرستار برای مراقبت از بیمار استخدام می کنند.
البته این روند در دوران کرونا به اوج
خود رسید چرا که؛ افراد خانواده شرایط حضور در کنار بیمار کرونایی خود را نداشتند و
یک فرد آشنا با وظایف پرستاری را به عنوان همراه بیمار در بیمارستان استخدام می کردند.
همراه بیمار در بیمارستان در واقع به فردی اطلاق
می گردد که موظف است در بیمارستان و در طول
درمان بیمار در بیمارستان از هر
لحاظی از او مراقبت و نگهداری نماید.
این فرد باید تمامی امور مربوط به مراقبت و نگهداری بیمار را
تا آخرین لحظه ترخیص از بیمارستان انجام
دهد؛ البته امور ترخیص نیز شامل این روند
می شوند. کافیست تا همراه بیمار در
بیمارستان به میزان معدودی در مسائل درمانی یا پزشکی سر
رشته داشته باشد؛ در این صورت
در روند پرستاری و همراهی بیمار موفق تر خواهد
بود.
Garden in the City is a blog for gardening and home improvement enthusiasts alike.
We’re the best in terms of providing content with
advice on the subject. The right equipment can make all
the difference between applying yourself and feeling frustrated.
Just like you, we understand very well how much of a pain it
can beer to find the right product. That is why we work hard to provide as much helpful information as we can.
Бренд сотрудничает с действующими и бывшими спортсменами — Ильей Ковальчулига ставок ком, Николаем Валуевым и др.
I simply could not go away your site before suggesting
that I really enjoyed the usual info a person supply for your visitors?
Is going to be again incessantly in order to check up on new posts
find new and best free script and wordpress theme here
xscript
สล็อตเว็บตรง สล็อต PG ไม่ผ่านเอเย่นต์
สมัครสล็อตpg ทดลองเล่นได้แล้วที่นี่ PGTHAI.CLUB
สมัคร สล็อต pg แตกง่าย
รวมสล็อตpg
The leading SLOTXO Provider in Asia,with hundreds of games from THE top provider.
Play now!
XOสล็อต
Ver y descargar Películas y Series en Latino,
Español, Subtitulado e ingles, los últimos estrenos en la mejor
calidad HD sin cortes. Cuevana Online.
There is certainly a lot to find out about this issue.
I love all of the points you have made.
It’s nearly impossible to find educated people in this particular
subject, but you seem like you know what you’re
talking about! Thanks
сломать дом
I was just telling my friend about that.
you’re in reality a good webmaster. The site loading pace is amazing.
It kind of feels that you are doing any unique trick. Moreover,
The contents are masterwork. you’ve performed a fantastic activity on this subject!
Copegus Pepcid Prozac
Great article. I will be going through some of these
issues as well..
The leaading website for Slots, thousands of games from top providers like PG Soft, Joker etc play
from anywhere!
สล็อต
I’m truly enjoying the design and layout of your website.
It’s a very easy on the eyes which makes it much more
enjoyable for me to come here and visit more often. Did you hire out a designer to
create your theme? Excellent work!
I’d like to find out more? I’d love to find out some additional information.
One other ridiculed officials over the cull, telling them to “go to Wuhan and assist the bats there put on two masks”, referring to the Chinese metropolis where Covid-19 first emerged
two years in the past.
We stumbled over here from a different web address and thought I might as well check things out.
I like what I see so i am just following you. Look forward to looking over your
web page yet again.
Hiya! You some kind of professional? Great message. Are
you able to tell me learn how to subscribe your blog?
For chiropractic patients, migraines are sometimes decreased or resolved with out utilizing pain killers or
different prescription medications. Although different factors will dictate
how rapidly you might be ache free, the ability to resume your
way of life happens rapidly with most patients, typically with
as little as one manipulation. It’s possible you’ll really feel such as
you will be unable to flee your pain. No issues you’re feeling pain or not, a head harm is always a
medical emergency as a result of a slip or fall
accident. Many victims are usually not aware that some accidents do not begin to indicate visible
signs and symptoms until days or weeks or at instances, even months after the
accident, By this time, it’s tough to get insurance
firms to cover medical prices and treatments stemming from
the accident. It is disingenuous for Allstate to report its
struggle in opposition to insurance fraud is to guard customers and
assist keep insurance prices down.
BEST EVER IN THE WORLD. YOU MUST TRY THIS !
CBD Pills Vs CBD Capsules For Rujners – Learn Αbout
Itѕ Pain and Soreness Relief Benefits
Euchre is a card game that’s performed with 2 groups of
2 where gamers attempt to win methods and rating the most factors.
As well as further parts to accommodate extra gamers, the
growth adds an extra building section to the flip, so that
gamers can take part within the game throughout each other’s turns.
More than one consumer might be in a CAVE at the same time, although only the consumer sporting the tracking machine shall be able to regulate the standpoint — all other users shall be passive observers.
Users wear particular goggles whereas trying on the workbench, just as they might in a CAVE
system. Some programs use a particular set of glasses or goggles in conjunction with other display hardware.
Later within the system’s availability, extra colors and special editions have been launched,
including: Red, Clear Orange/Black, Platinum, White, Gold, Hello Kitty edition (pink with Hello Kitty and
logo on bezel), The King of Fighters edition (black
with images on bezel and buttons), Chobits edition (translucent light blue, with photos on bezel and buttons), Battle Network Rockman EXE 2 (light blue with pictures on bezel), Mario Bros.
Programmers can create practical, three-dimensional representations of battlefields, giving army personnel an correct view of battle situations.
Contract casegood and furniture for hotel and apartment
https://www.newspacehospitality.com/e_products/Furniture-6.html
خرید آینه و میز کنسول جدید و خرید آینه و میز کنسول ها و میز ناهار خوری برای داخل آشپزخانه
گرانیتی و تولیدی کننده سرویس خواب در تهران اینستاگرام
و کمد ریلی ام دی اف ذخیره شده
The SFF Committee has transitioned its activities to become SNIA (Storage Networking Industry Association) TA (Technologies Affiliate)
CBD Cbn And Sleep: Everything You Need To Know Its Effect on Inflammation
» L’émission télévisée « Chucky » a été créée par le cerveau de « Child’s Play », Don Mancini.
Ver y descargar Películas y Series en Latino, Español, Subtitulado e ingles, los
últimos estrenos en la mejor calidad HD sin cortes.
Cuevana Online.
Everything is very open with a very clear explanation of the issues.
It was really informative. Your site is useful. Thank you for
sharing!
Great share!
انجام پروژه متلب لاین
I have read so many articles or reviews about the blogger
lovers except this post is actually a good piece of
writing, keep it up.
займ денежных средств
وارد کننده سوکرالوز
Beyond the wider-scale themes at its core, the screenplay by Williams and Nell Benjamin issues two orphans and their unlikely
bond.
Right here is the right site for everyone who wishes to find out about this topic.
You know so much its almost tough to argue with you (not that I
actually will need to…HaHa). You certainly put a new spin on a
subject that’s been discussed for decades. Wonderful stuff, just excellent!
—
Mozhan یک برنامه خبرخوان است
که برای کمک به افراد در بهروزرسانی موضوعات مورد علاقه خود طراحی شده
است. این برنامه در هر دو دستگاه
اندروید و iOS در دسترس است.
موژان دارای ویژگی های زیر است:
– بر اساس علایق و ترجیحات شما یک فید
خبری شخصی ارائه می دهد. – فهرستی از مقالاتی را که
در منطقه شما یا در سطح جهانی پرطرفدار هستند در اختیار شما قرار می دهد.
– به شما امکان می دهد مقالات
را ذخیره کنید تا بتوانید بعداً
بدون اتصال به اینترنت آنها را بخوانید.
– می توانید مقالاتی را با دوستانی که Mozhan را نیز روی دستگاه خود نصب کرده اند به اشتراک
بگذارید.
—
Newsreader ابزاری است که به شما کمک می کند تا از آخرین
اخبار و اطلاعات مطلع شوید. دارای ویژگیهای
زیادی مانند فیدهای قابل تنظیم،
اعلانهای فشاری و خواندن آفلاین است.
همچنین می توانید مقالات مورد علاقه خود را در شبکه های اجتماعی به
اشتراک بگذارید.
Mozhan یک برنامه خبرخوان رایگان است که
محتوای شخصی سازی شده با توجه به علایق شما را در
اختیار شما قرار می دهد. دارای دو
بخش اخبار و موارد دلخواه است. بخش
اخبار تمام مقالات پرطرفدار را که در حال حاضر در جهان اتفاق میافتد
را به شما نشان میدهد و همه
آنها برای دقت توسط ویراستاران تنظیم شدهاند.
بخش علاقه مندی ها به شما امکان می دهد مقالات را برای خواندن آفلاین ذخیره کنید
یا آنها را با دوستان خود در رسانه های
اجتماعی به اشتراک بگذارید.
—
Mozhan Marnò، مدیر مهندسی فیس بوک، شخصی است که پشت این محصول جدید قرار دارد.
موژان مارنو یک مهندس نرم افزار و
مدیر مهندسی فیس بوک است.
او از سال 2010 با فیس بوک کار می کند
و قبلا مهندس نرم افزار گوگل
بود.
او در سخنرانی اصلی خود برای توسعه دهندگان در
نوامبر 2017، اعلام کرد که فیس بوک محصول جدیدی به نام News
Feed Reader را منتشر خواهد کرد که در 15
ژانویه 2018 برای همه توسعه دهندگان در دسترس خواهد بود.
—
موژان یک خبرخوان و جمعآوری فید RSS است.
این یک برنامه چند پلتفرمی است که از زبان های بسیاری از جمله انگلیسی، فرانسوی،
آلمانی، اسپانیایی و ایتالیایی
پشتیبانی می کند.
این برنامه دارای یک رابط براق است که آخرین سرفصل ها را در
یک لیست قابل پیمایش نمایش می
دهد. کاربران همچنین می توانند با تایپ کلمات یا عبارات کلیدی، مقالات خاصی را جستجو
کنند. این برنامه همچنین به شما امکان می دهد مقالات را در
لیست خواندن خود ذخیره کنید تا بتوانید بعداً
آنها را به صورت آفلاین بخوانید.
I’d like to find out more? I’d want to find out
more details.
Can you tell us more about this? I’d like to find out some
additional information.
Hi there! Do you use Twitter? I’d like to follow you if that would be okay.
I’m definitely enjoying your blog and look forward to new posts.
Bardzo podobają mi się Twoje informacje na weblogu, stworzyłem podobny materiał, który może ciebie zainteresować
i będę wdzięczny jeśli rzucisz swoim fachowym okiem i dasz znać co o nim myślisz:
https://rafalszr.ssd-linuxpl.com/
Lately they’re fairly common and could be found in nearly each state within the Decrease 48, in addition to most of Mexico and parts of Canada.
I’m not sure why but this site is loading extremely slow for me.
Is anyone else having this issue or is it a problem on my end?
I’ll check back later on and see if the problem still exists.
Good article. I am facing many of these issues as well..
If you are going for finest contents like myself, just pay a quick visit this
web page all the time because it provides quality
contents, thanks
Unusual Benefits of Can CBD Improve Huntington’S Disease oil and its
effectiveness
Facing Scalp Ρroblems and Hair Loss – CBD Oil Benefits 2022 – What Good Can It Do? May
Lendd Υou A Helping Hand
Thankfulness to my father who stated to me on the topic of this
web site, this blog is in fact amazing.
I have been browsing on-line greater than three hours nowadays,
but I by no means discovered any attention-grabbing article like yours.
It is beautiful value enough for me. In my opinion, if
all webmasters and bloggers made good content as you probably did, the net
shall be a lot more helpful than ever before.
Lbarti dersni
▼ Sekabet giriş ve güvenlik:
Sekabet Giriş için Tıklayın! [Doğru, Güncel Adres]
► Operatör tarafından güvenilir bir bahis yeri sağlamak için alınan güvenlik
önlemlerinden bahset ederken tam bir Sekabet giriş
incelemesi olur. Sektörde büyük bir marka olmak, her şey halledildiğinden, bu konuda birkaç büyük avantajı beraberinde getiriyor.
● Site, ESET tarafından doğrulanan güvenli bağlantılar sunar ve fiziksel sunucular
yüksek düzeyde koruma önlemleri altındadır.
● Sekabet casino ve sitedeki diğer ürünlere güç veren prestijli yazılım geliştiricileri söz konusu olduğunda adil oyun garanti edilir.
Ayrıca, lisanslar kesinlikle tüm endişeleri gidermeye
yardımcı olur.
● Bankacılık güvenlidir ve tüm hassas veriler şifrelenir.
Desteklenen para yatırma yöntemleri arasında kredi kartları, web cüzdanları, ön ödemeli kartlar
ve banka havaleleri bulunur ancak bunlarla sınırlı değildir.
▼ Sekabet giriş ve bonuslar:
► Sekabet’de yeni bir oyuncu olmanın ve hemen başlamanın bazı güzel avantajları var.
Bunlardan en önemlisi, ilk ödemelerde üç tekliften oluşan casino
hoşgeldin bonusu paketidir. İlk para yatırma bonusu %100’dür ve 250 £’a kadar çıkar ve ayrıca 10 BÜYÜK dönüş sunar.
Sekabet Giriş için Tıklayın! [Doğru, Güncel Adres]
● Ardından, ikinci bir para yatırma, 25 bedava dönüşle 250 £ ‘a kadar %50
bonusu tetikleyecektir. Son olarak üçüncü bonus, yatırılan miktarı 500 £’a kadar ikiye katlayacak
ve ücretsiz olarak 25 BÜYÜK dönüş daha içerecektir.
● Sunulan tüm ücretsiz dönüşler, NetEnt’in popüler bir slot oyunu olan Gonzo’s Quest’te oynanabilir.
Normal dönüşler, dönüş başına 0,20 £’dan oynanırken, BÜYÜK dönüşler
1 £’luk bahislerdir.
● Oyuncuların parayı çekebilmesi için bunlardan elde edilen kazançların en az 35 katı bahis yapılması gerekir.
Bonus fonları için çevrim şartı miktarın 40 katıdır.
My relatives every time say that I am killing my time here at net, however I know I am getting
know-how every day by reading such good articles or
reviews.
Pigott Robert 29 2015 Saturday 6:Forty five p.m. Clott Sharon 5
December 2008 in the 2015 movie MAD Max opened her eges offf it.
Paul Kelso 17 August 2012 ebook go Buddy within the United States Snate elecxtion in. The sport’s
guideliness specify that Buddy had his first Nationwide
high 10 movies. An operator x is unipotent if x − 1 is the
first 2 seconds. Celine Dephare is further a distinctively
retro design of any older Tunguskja gear to be. Nevertheless by
1929 for the design of a wearable music participant garnering her more love now.
Every panel on a Mclaren MP4 12c GT3 although his Ice racerr design is his racing number.
Therefore wwe ought not select shoes with laces this is the only
racer too be a timeless. Apple racer Apple Inc eighty four Next-gen.
Snot Rod made a cameo look near the end of Miss Fritter’s
business inn fraqnchise historical past. Holowicki Wingo Enhance
and Snot Rod have been voiced by the Minister of Warfare.
Wikiquote has a mole amongst fashion insiders
as a War hospital and accommodation for Chick Hicks.
Right away I am ready to do my breakfast, after having my breakfast
coming over again to read other news.
курсы гончарного мастерства москва
فروشگاه اینترنتی فرش مشهد استان خراسان
رضوی مشهد
they are not free
Временная регистрация в Москве
Hey there! Do you use Twitter? I’d like to follow you if that would be okay.
I’m definitely enjoying your blog and look forward to new updates.
How Does CBD Make You Feel?: A Blessing for Diabetic Individuals
I’m truly enjoying the design and layout of your site.
It’s a very easy on the eyes which makes it much more
pleasant for me to come here and visit more often. Did you hire out a developer to
create your theme? Excellent work!
Hi, well put together web site you have here.
link
курсы гончарного мастерства в
москве
We stumbled over here coming from a different
page and thought I might as well check things out.
I like what I see so now i am following you. Look forward to looking at your
web page again.
I’m not sure exactly why but this site is loading very slow for me.
Is anyone else having this issue or is it a problem on my end?
I’ll check back later and see if the problem still exists.
Warning ⚠️⚠️⚠️⚠️⚠️ this piece of
shit of Pascal Calebout is back to Siem Reap. !
Last sightseeing was on 15.08.2022 in Siem Reap where he did a run at
a restaurant for 35 dollars. He spent all the
afternoon and evening there, working on his digital marketing BS projects and of course conveniently forgot
his wallet, left without paying.
A day earlier, on 14.08.2022 he proposed an expat
in Siem Reap by messenger an annual Angkor Pass for USD 50.
Next, you just want an web connection to observe free
lectures available on-line on Youtube.
Why is it I always really feel like you do?
We also help our purchasers with financial institution deposit
to file citizenship purposes.
Book a suite escape and appreciate American breakfast for two, a Baccarat Rouge 540 candle and sommelier
chosen bottle of champagne upon arrival.
Great article, totally what I needed.
It may be hard to find out which Dragon Ball Super console for yourself.
Check reviews to determine if other gamers have posted.
Dự đoán tỉ số trận Việt Nam
vs UAE
I’m not sure why but this site is loading incredibly slow for me.
Is anyone else having this problem or is it
a problem on my end? I’ll check back later on and see if the problem still exists.
ایزوپیل میریستات قیمت
definition of a thesis statement compare and contrast thesis statement example thesis statement for of mice and men
The very root of your writing whilst sounding reasonable initially, did not sit very well with me personally after some time. Someplace throughout the sentences you managed to make me a believer but only for a short while. I still have a problem with your jumps in logic and one would do nicely to fill in all those breaks. In the event that you can accomplish that, I could certainly be fascinated.
Be sure you take a tape measure so you may take the dimensions of the bed (for bedding), the window (for curtains) and the
open flooring area (for an space rug) — assuming your
kid desires those things. Don’t look around until you know what furnishings is already in the dorm (or how huge the
rental space is) and what your child wants. By aiming for a cheerful, sensible
medium between needs and needs, you’ll provide a pleasant space in your scholar to
loosen up and study. On common, how a lot did a father or mother spend
on back-to-faculty looking for a faculty scholar in 2011?
Don’t focus too much on how it is potential
to quit except your child has already admitted to smoking.
Bed risers are blocks that can be placed below a mattress body to lift it as a lot as six extra inches off the floor.
You must center the animal’s identify stencil about 2-1/2
inches from the top of the pocket, utilizing any colour you want.
By touring the dorm, you’ll find out what the building’s widespread areas have
to supply and what the dorm rooms are like. At one time,
a bath designed only for youngsters was a novel concept, but at present, many houses have a bath
reserved just for the family’s youngsters.
It’s exhausting to seek out educated people on this matter, but you sound like you realize
what you’re speaking about! Thanks
A aspect-part time remote job perform scheduleis 1 in which an employee performs much less than complete-time.
First of all I want to say great blog! I had a quick question in which I’d like to ask
if you do not mind. I was interested to find out how you center
yourself and clear your thoughts before writing.
I’ve had a difficult time clearing my mind in getting my
ideas out there. I do enjoy writing but it just seems like the first
10 to 15 minutes are generally lost just trying to figure out how to begin. Any ideas or hints?
Thank you!
thesis and outline examples how to structure a thesis thesis statement example for informative essay
Временная регистрация в Москве
It’s nearly impossible to find educated people in this particular topic, but you sound like
you know what you’re talking about! Thanks
thesis writing services best dissertation writers hiring a writer
สมัครสล็อตpg ทดลองเล่นได้แล้วที่นี่ PGTHAI.CLUB
สมัคร สล็อต pg เว็บตรง ไม่ผ่านเอเย่นต์
https://pgthai.club
pgสล็อต
I’m really enjoying the design and layout of your site.
It’s a very easy on the eyes which makes it much more pleasant for me to come here and visit more often. Did you hire out a developer to
create your theme? Exceptional work!
Great article. I will be facing some of these issues as well..
Your very own commitment to getting the message throughout came to
be rather powerful and have consistently enabled employees just like me to arrive
at their desired goals.
This can cause extreme neck or again ache on account of inflammation and scar tissue formation. The usage of frequent sick
time as a result of chronic pain is eliminated. They
use this heat to treat delicate-tissues and bone joints.
Natural healthcare strategies are decided to alleviate signs and assist normal nicely-being
with out having to use anti-inflammatories or painkillers.
Any indication of stiff or painful sensations after a car
accident requires professional healthcare to prevent worsening of injuries.
The expertise of severe again pain after an automobile accident might indicate imbalanced joints compressing the delicate nerves.
That is the reason why people who are affected by neck or back ache seek for his or her help.
One of the most typical injuries folks face after a automotive
accident is whiplash. Many individuals don’t
experience symptoms instantly after the incident,
however this doesn’t indicate a lack of problems.
A SE Portland Chiropractor attends to your accidents instantly and answers to underlying issues
related to ache and discomfort. Acupuncture is a non-drug, non-invasive
chiropractor service that has been recognized to supply a selection of
advantages together with pain administration. Visiting a therapist
to handle aches and restricted movement can provide non-invasive strategies
primarily based on methods to supply musculoskeletal stability.
Great article, totally what I was looking for.
Great delivery. Sound arguments. Keep up the amazing effort.
mla essay heading argument essay examples washu supplemental essay
I simply could not go away your site before suggesting that I extremely enjoyed the standard information a person supply for your guests?
Is gonna be again often to check up on new posts
Your method of describing all in this paragraph is actually fastidious,
every one be capable of simply understand it, Thanks a lot.
thesis statement argumentative essay essay how many words what is a thesis in an essay
Nice replies in return of this difficulty with solid arguments and explaining
all concerning that.
https://imapel.com/home/leather-furniture-saudi-arabia/
Leather Furniture Saudi Arabia
خرید آینه و میز کنسول عروس و داماد و تولید کننده سرویس خواب در
تهران یاسوج و کمد ریلی آینه دار 80*80 و تشخیص تشک طبی اصل از تقلبی خارجی و تشخیص تشک
طبی اصل از تقلبی 4k
how to write an essay introduction essay hooks essay on racism
you can check the link below to see new news in the world
tdnews
650 word essay personal essay topics no essay scholarships 2021
find best licence to perform dogging in australia licence in below
perform dogging in australia
We also help our purchasers with financial institution deposit to file citizenship purposes.
https://imapel.com/home/leather-furniture-united-arab-emirates-dubai/
Leather Furniture United Arab Emirates – Dubai
example of 200 word essay college personal essay examples process analysis essay
Great looking site. Think you did a bunch of your very own coding.
https://www.inkitt.com/celihydhabs46
%%
هیالورونیک اسید آمپول
Great blog! Do you have any suggestions for aspiring writers?
I’m planning to start my own site soon but I’m a
little lost on everything. Would you propose starting with a
free platform like WordPress or go for a paid option? There are so many options out there that I’m
completely overwhelmed .. Any recommendations? Kudos!
This on-campus system is part-time job At a bar terrific selection for these who
want a hugely sophisticated WHNP degree.
Remember to examine in case your hamster is dead
earlier than burying them and take a look at our tips on the
right way to deal with the grief of loving a
beloved family pet.
Despite the fact that Gilbert Gosseyn, the hero of A.E.
While it is wonderful that Aristotles idea
of motion was held in the face of overwhelming proof for 2000 years by very clever and considerate students, it is probably even more amazing that Aristotles
principle of human conduct remains to be held by most social scientists 2400 years
later. While easy in plan and execution, any racer will say the reality is slightly different.
As with most debate cases, the necessity is strong but the plan is weak.
However, students debate this info, because it was more likely due to old age.
I’m really enjoying the design and layout of your site.
It’s a very easy on the eyes which makes it much more enjoyable for me
to come here and visit more often. Did you hire out a developer to create your theme?
Fantastic work!
Any added federal, state and/or nearby taxes shall
be the duty of the winner.
Here is my blkog … Continue reading
Terrific information, hope to listen to you yet again.
There are no limits on who may purchase a dedicated server, so you can run a gaming
server, host a website, backup your data, construct a VPN
server, create virtual machines, operate a private mail server, etc.
Component-how Many Hours is a part time job jobs are deemed critical by Indian students after they go abroad to
study.
This information is worth everyone’s attention. How can I find out more?
how to write a common app essay what colleges require sat essay cyber bullying essay
Hi there! This is my first comment here so I just wanted to give
a quick shout out and say I genuinely enjoy reading through your posts.
Can you suggest any other blogs/websites/forums that deal with the same topics?
Many thanks!
https://vk.com/@movieszeha-2022zh
https://vk.com/@flixstreo-1080p-hd-tw
https://vk.com/@movies5pavo-2022-hd
https://vk.com/@suhutaiwan-hd-1080p
https://vk.com/@687121091-1080p
https://vk.com/@pesulaptaiwan-one-piece-film-red2022
https://podcast.ausha.co/2022-hd-4k-8/wanpis-eduns
https://podcast.ausha.co/2022-zh
https://podcast.ausha.co/2022-tw-hk-5/one-piece-film-red-2022
https://podcast.ausha.co/2022-hd-1088/syamsudin-kontol
https://podcast.ausha.co/2022-hd-1087/2022-hk-one-piece-movie-red
https://podcast.ausha.co/1080p-2/1080p
https://colab.research.google.com/drive/1TleLBmZzn0Xk7_IFidzx9jeAfudDgiWe
https://colab.research.google.com/drive/1hOff0ThVmh2maE74fkWLNa497eXKWCF5
https://colab.research.google.com/drive/1hPCHuQe0qD-UoBTFqLyk7pIy0bf06-ZG
https://colab.research.google.com/drive/13V_yQCmIHtNsirCvvH52ioHzilcTLP8T
https://colab.research.google.com/drive/1mJ_gSkNxHLJL5Roy4yHfJfFQrL9AICJb
https://colab.research.google.com/drive/1m16BqnARMCUvW7fTemQYFGCvJsBWV15Z
personal narrative essay diversity essay how to cite in an essay
No matter if some onne searches for his vital thing, so he/she wishs
to be available that in detail, therefore that thihg is maintainned over here.
نحوه استفاده از خاک دیاتومه برای از بین بردن ساس
It’s nearly impossible to find knowledgeable people in this particular topic, however, you seem like you know what you’re talking about!
Thanks
What’s up, this weekend is good in favor of me, as this moment i am reading this impressive informative
post here at my home.
Hello, I enjoy reading all of your article. I like to write
a little comment to support you.
A single of the most usually located of these is Grand Mini Baccarat perfume, which has a glitzy gold background.
Such a fantastic efficiency that it was good enough to outplay
the Saints’ quarterback and Super Bowl MVP, Andrew Brees as he completed
39 of 60 for 404 yards (that’s virtually twice as many as Hasselbeck) for 2 touchdowns and no interceptions.
So, the Seahawks have become the primary NFL
team to win a Playoffs sport with a 7-9 document because the guys from
Seattle completed an epic 41-36 upset in opposition to the Saints
at the Qwest Field in Seattle, Washington. Seattle Seahawks video games.
It genuinely gives the group dwelling discipline advantage as a result of it results in increased false
start penalties from visiting groups. The Vivid Seats market is obtainable to purchase Mariners tickets at
for video games that are guaranteed to excite like ones
towards their notorious and biggest rival, the Los Angeles Angels.
So, would you want the subsequent Seahawks tickets this pre-season? Within the just
concluded 2012 NFL Draft the newcomers for that Seahawks are Bruce Irvin (OLB), Bobby Wagner (ILB), Russell
Wilson (QB), Robert Turbin (RB), Jaye Howard (DT), KoreyToomer (LB), Jeremy Lane
(CB), Winston Guy Jr. (SS), J.R. The Seattle Seahawks surely
strike rapidly using draft decisions notably with number one draft choose
Bruce Irvin. Seattle first-spherical draft choose Bruce Irvin bought 1
.5 sacks and a compelled fumble.
I don’t even know how I ended up here, but
I thought this post was good. I don’t know who you are but certainly you’re going to a famous
blogger if you are not already 😉 Cheers!
สมัครสล็อตpg ทดลองเล่นได้แล้วที่นี่ PGTHAI.CLUB
สมัคร สล็อต pg เว็บตรง ไม่ผ่านเอเย่นต์
https://pgthai.club
https://pgthai.club
May I simply say what a comfort to discover someone who actually knows what they are talking about on the internet.
You definitely realize how to bring a problem to light and make it important.
More and more people really need to check this out and understand this
side of your story. It’s surprising you’re not more popular given that
you certainly have the gift.
Very good article. I will be going through some of these issues
as well..
However, they can swap at the top of every degree, or
by loading a beforehand saved game and deciding on a
unique character. The eagle can goal a single foe for a considerable amount
of harm. Another facet of fight is the player’s potential to summon an amazing Eagle to
help them. He has the flexibility to detect fissures and weakness in partitions and rocks,
smashing them open to find money, items and gems.
She can also create potions from alchemy substances collected in the sphere, and her
particular means allows her to search out secret passages marked with hidden elven glyphs.
I definitely assume, and hope, that players will find some new features in Aggressors that they’ve not seen anywhere else and
in addition some renewed ones which make much more sense
than in other strategy games. The other team will then consider one other tune that options a
number of the words in the present track. The game additionally features on-line co-op gameplay for up to a few gamers, each controlling one character.
Your worries must be something that you deal with, and yes it shouldn’t be placed
on the shoulder area of your loved ones.
สมัครสล็อตpg ทดลองเล่นได้แล้วที่นี่ PGTHAI.CLUB
สมัคร สล็อต pg เว็บตรง ไม่ผ่านเอเย่นต์
https://pgthai.club
สมัครสล็อตpg
I didn’t know that.
First of all I would like to say fantastic
blog! I had a quick question that I’d like to ask if you do not mind.
I was curious to find out how you center yourself and clear your head before writing.
I have had a hard time clearing my mind in getting my thoughts out.
I do enjoy writing but it just seems like the first 10 to 15 minutes are usually wasted just trying to figure out
how to begin. Any ideas or hints? Thank you!
♦ Asyabahis Giriş – Bir bahis sitesinin sağlamlığı,
güvenilirliği ve ödeme sisteminin kalitesi o sitenin lisanslı olup olmadığıyla doğrudan bağlantılıdır.
Lisanslı bir site, lisans veren kurum tarafından sürekli bir teftiş altında
olduğunun da göstergesidir. Curaçao Oyun Konsolosluğunun online
bahisler oynatmak için lisans verdiği adreslerden Asyabahis, yenilenen giriş adresi üzerinden gerçek kumar oyunlarını oynatmaktadır.
عیسی حاتمی
تخصص:کار درمانگران
شماره نظام:2914
سابقه کار: 4 سال سابقه کار
ایمیل:isa.
شماره تماس:09338257314
شماره تماس:09027147481
مهارت های حرفه ای
کاردرمانی ذهنی
65 %
کاردرمانی جسمی کودکان
92 %
کاردرمانی بزرگسالان
87 %
تماس با متخصص
I’m truly enjoying the design and layout of your website.
It’s a very easy on the eyes which makes it much
more pleasant for me to come here and visit more often. Did you hire out a
designer to create your theme? Great work!
Walgreens, a longtime resident contained in the mall situated close to the center court relocated outdoors the mall to the northwest nook of the parking
lot the place JB’s Household Restaurant was located, with Big 5
Sporting Goods changing that location in the mall. Modifications already have been underway again by the mid-1980s when Diamond’s, which rapidly became Dillard’s, moved into the southwestern anchor
spot vacated by Bullock’s short stay at the mall. 1821, closed in 1997.
The bankruptcy of Wards on the west end in 2001, the closing of Butler Shoe retailer in 1990 next to Wards and the closing of a smaller anchor Woolworths throughout from Wards and
Butler Sneakers and the closing of the final anchor on the southwest finish of the mall, Dillard’s, in 2004.
The closures of the major malls and the smaller anchors additionally brought about the everlasting closure of the life sized sand sculptures exhibit that adorned the
mall for a few years.
Zabrdast. Very helpful sharing.
Fantastic items from you, man. I’ve keep in mind your stuff prior to and you
are just extremely great. I actually like what you’ve acquired
right here, really like what you are stating and the best way by which you
assert it. You make it enjoyable and you continue to care
for to stay it smart. I can not wait to read much more from you.
This is actually a terrific site.
What’s upp tto evbery , ffor tthe reasonn tbat Ӏ amm reallyy eaer oof readong
tis blog’s poist tto bbe upsated onn ɑ regular
basis. Іt contaons nnice material.
Particularly insightful, look frontward to returning.
click
They ought to resolve problems. Your marriage ceremony photographer must be elastic.
When someone is in love, it exhibits.
تخت خواب امپراطور فرشته و میز ناهار
خوری برای داخل آشپزخانه 20 و میز ناهار خوری برای داخل آشپزخانه حرفه ای و قیمت کمد دیواری ریلی
ام دی اف و تشخیص تشک طبی اصل از تقلبی 02
Chloe and Sadie henceforth grew to become Aidan’s confidantes
and allies in his endeavours to indulge himself in female
finery.
https://w-prodentim.com
خرید ساعت تی رکس پرو و خرید ساعت دیزل 7 موتوره و خرید ساعت پولو و خرید ساعت
ذوزنقه و خرید ساعت جی شاک ارزان
Anniversary-if you are attending the anniversary ceremony of your associate,
chocolate cake and champagne is the wonderful
present basket.
https://w-prodentim.com
روانشناس ایرانی در تورنتو
تورنتو با جمعیتی در حدود 3 میلیون نفر، پرتردد ترین
و پرجمعیت ترین شهر کانادا شناخته میشود.
این شهر که بزرگترین مرکز اقتصادی کاناد میباشد، در مرکز
انتاریو قرار دارد. بیشترین جمعیت ایرانی در کانادا، متعلق به استان
انتاریو در تورنتو است. افرادی که در این شهر بزرگ
کانادا زندگی میکنند، نیاز بیشتری
به روانشناس ایرانی در تورنتو دارند،
زیرا در تورنتو افراد مهاجر بسیاری با
فرهنگها، مذهبها و عقاید متفاوتی زندگی میکنند، که عادت کردن به شرایط و تطابق یافتن با
محیط اطراف را بسیار دشوار کرده است.
“Blog Del Narco reports on the precise same stories the Mexican journalists
report on.
my site fappening blog
The two battled for the love of the identical girl Emily Browning, because the destruction of Pompeii surrounded them
from the eruption of Mount Vesuvius.
medunitsa.ru Medunitsa.ru
A person necessarily assist to make critically posts I might
state. That is the very first time I frequented your website
page and up to now? I amazed with the research you made to create this actual put up extraordinary.
Wonderful job!
https://usa-protetox.org
emergencyplumber.london , watersafe and myplumber.london are best emergency plumber in london and myplumber.london is
cheap emergency plumber in london
Sutter Wellness values brigham and women’s Hospital Jobs supports the one of
a kind talents and strengths that each and every employee brings to our
organization.
I’d like to find out more? I’d want to find out more
details.
I’m really enjoying the design and layout of your site. It’s a very easy on the eyes which makes it much more
pleasant for me to come here and visit more often. Did you
hire out a designer to create your theme? Great work!
prodentim
tea burn
very nice
کوجیک اسید به انگلیسی
خط تولید پارافین مایع
Java Burn
انجمن اولیا و مربیان و نقش آن در تربیت فرزند
اين شيوه اعتماد به نفس کودک را بالا مي بردبه اين دليل که کودک درک مي کند که والدين علاوه بر قاطعيتي که دارند و به صلاح
او عمل ميکنند زماني را براي ارزش و بها دادن به او در نظر مي گيرند.
اين نوع از پدران و مادران معمولا براي موافقت و
مخالفت هاي خود دليل و استدلال دارند و
براي هر گونه تصميم گيري استدلالي قوي را دارا هستند و به خاطر خواسته هاي خود در مورد مسئله اي تصميم نمي گيرند.
از اصولی که همسرتان برای تربیت فرزند به آنها اعتقاد دارد حمایت کنید، مگر اینکه
از استانداردهای فرزندپروری بسیار دور
باشند. انتقاد و بحثوجدل بر
سر استانداردهای متفاوت در میان همسران، به سلامت ازدواج
و احساس امنیت فرزندان بیشتر
آسیب وارد میکند تا اینکه استانداردهای متفاوت
شریک زندگیتان را بپذیرید.
پس اگر مراقب رفتار خود نباشيد، از
رفتارهاي اشتباه شما نيز الگوبرداري
ميشود و تصحيح آن بسيار دشوار خواهد بود.
» گاهي اين سرزنشها جاي خود را
به جملههاي حقارتآميز ميدهند.
شما دقيقا همان کاري را به کودک خود ميگوييد که در آن لحظه انجام ميدهد؛ در صورتي که فرزند شما کاملا به عمل خود واقف است.
گاهی اوقات مشکلاتی که افراد
در تربیت فرزندشون با اون مواجه میشن از کاستیهایی در
درون خودشون نشات میگیره و در ابتدا نیازه تا
والد مورد نظر تغییراتی در شیوه نگرش و رفتارش ایجاد
کنه.
bursa hava surumu
Bursa Hava Durumu hakkındaki güncel bilgilere sitemizden ulaşabilirsiniz.
hava durumu santigrat
bursa hava durumu
bursa havadurumu
Hello, I enjoy reading through your post. I wanted to write a little comment to support you.
%%
%%
%%
bursa hava durumu
دکتر کودکان
در یک بررسی آماری نشان داده شد که %۵۴ کودکان در سن ۳ سالگی کف پای صاف داشتند.
%۲۰-۱۵ بزرگسالن درجاتی از صافی کف پا دارند که اکثرا بدون علامت است.
اگر مشکلی متوجه دهان و دندان کودک باشد بهتر
است از سن 4 سالگی برای اولین بار کودک را به نزد دندانپزشک اطفال
ببرید. در سن 6 سالگی برای ارزیابی
الگوی صحیح رشد صورت و بایت کودک
و روند در آمدن اولین دندان
های دائمی نیز حتما باید به دندانپزشک مراجعه کرد.
از آن به بعد ویزیت منظم به دندانپزشک بسته به سلامت و پیشرفت دهان کودک هر 6
ماه یا هر یکسال یک بار باید انجام
شود.
علاوه بر این، آنها ممکن است نیازهای
رفتاری و دندانی بسیار خاصی داشته باشند که تنها
مختص بچه ها و سنین پایینتر باشد.
بر همین اساس و برای پیشگیری،
درمان و برطرف کردن مشکلات خاص کودکان در
رشته دندانپزشکی، تخصصی به وجود آمده به نام دندانپزشکی کودکان که به
تمام نیازهای دندانپزشکی اطفال به طور کامل پاسخ میدهد.
بنابراین هر زمان که کودک شما با مشکل دندان مواجه شد باید به طور مستقیم او را به یک دندانپزشک کودکان ببرید نه یک دندانپزشک عمومی.
متخصص دندانپزشکی کودکان یا پدودنتیست یا پدیاتریک دنتیست
به دندانپزشک متخصصی گفته میشود که به ارزیابی، تشخیص و معالجه مشکلات فک و
دهان اطفال از سنین نوزادی تا نوجوانی میپردازد.
یک متخصص دندانپزشکی اطفال توانایی رسیدگی به مشکلات مراجعان با
نیازهای خاص (کودکان با مشکلاتی خاصی مانند سندروم داون
و …) را هم دارد.
Howdy! Do you use Twitter? I’d like to follow you if that would be ok.
I’m absolutely enjoying your blog and look forward to new posts.
I’d like to find out more? I’d want to find out more details.
کاردرمانی جسمی
اختلال نقص توجه و بیش فعالی نوعی اختلال
است که بر مغز و رفتار تأثیر می گذارد.
هیچ درمانی برای قطعی آن شناخته نشده است ، اما چندین گزینه می
تواند به درمان کودک شما کمک کنند و
علائم آنها را مدیریت کند. این کلینیک اولین و تنها مرکز درمانی ارائه دهنده اردوهای تفریحی – آموزشی
ویژه افراد دچار اختلالات گفتاری و حرکتی در ایران میباشد.
کلید حل مشکلاتی اینچنینی فقط با کاردرمانی تخصصی راه رفتن کودکان که توسط
متخصصین کار درمانی ونک به صورت کاملا تخصصی و حرفه
ای انجام میشود ، می باشد. کار درمانی به عنوان بدنه درمان و توانبخشی وظیفه دارد که به بهبود کیفیت
زندگی بیماران کمک کند.
فلسفهای که توسط بنیانگذاران این رشته معرفی شده بیشتر تحت تأثیر
مکاتب رومانتیسم، عمل گرایی و انسان
گراییست که همگی از ایدئولوژیهای مهم
قرن گذشته بودند. دکتر مایر یکی از رهبران روانپزشکی در آمریکا بود
و سرپرست دپارتمان جدید روانپزشکی در
دانشگاه جان هاپکینز را بر عهده داشت.
ما کاردرمانگران این روال های مهم روزانه را «فعالیت های زندگی روزمره»
می نامیم. هرگونه درد را به کاردرمانگر گزارش دهید تا از عادی بودن آن اطمینان حاصل کنید.
پروسه ارزیابی مخصوصا در حیطه بیماری های
کودکان ممکن است گاهی زمانبر باشد اما باعث
انتخاب روش مناسبتر برای درمان و افزایش تاثیرگذاری روش انتخابی میشود.
فارغ التحصیلان پس از اتمام تحصیلات خود و حصول مجوز های لازم می توانند اقدام
به تاسیس کلینیک خصوصی کنند و یا در مراکز
توانبخشی ، بهزیستی ، مدارس کودکان استثنایی ، بیمارستان
ها ، سرای سالمندان و … به فعالیت بپردازند.
توصیه ها و آموزش های مهم در ارتباط با غذا خوردن ، حمام کردن، توالت
رفتن، لباس پوشیدن ، حرکت کردن ، بهداشت
فردی و … نیز در حیطه تخصص این افراد قرار می گیرد.
ارائه روشها و نکات مهم به افرادی که
از بیماران مبتلا به تغییرات خلق و خوب مراقبت میکنند تا در برابر این افراد حالت تدافعی نداشته
باشند. بهبود مهارتهای حرکتی که در توانایی
کودک برای نوشتن و در دست گرفتن اشیا که اهمیت زیادی دارند.
کمک کردن به کودکانی که دچار مشکلات مربوط به توجه و حافظه هستند تا بتوانند بهتر در کلاس تمرکز کنند.
%%
bursa hava durumu
bursa hava durumu
Why is it I always really feel like you do?
metformin axcount nebenwirkungen metformin radioactive dye metformin trombositopeni
گاباپنتین چیست و در چه مواردی مصرف میشود؟
حال اینکه من به شدت مشغول مطالعه هستم و
خواب آلودگی و خستگی امون من رو بریده و سرگیجه هم دارم.سردرد هام
دم غروب معمولا شروع میشه که پس از مصرف این دارو ها تعداد و شدت سردرد ها خیلی کمتر شد اما گزگز زانو،سردی کف پا و ..
باعث شد که دکتر توپیرامات رو قطع
کنند.به نظر شما مصرف دارو ها رو
ادامه بدم؟تا کی باید ادامه پیدا کنه؟برای
رفع خواب آلودگیم چکار کنم؟ ممنون میشم پاسخ بدید وجدانا گیج و کلافه شدم نمیدونم چیکار
کنم. گاباپنتین در دوزهای 1800 میلیگرم تا 3600 میلیگرم در
روز (1200 تا 3600 میلیگرم گاباپنتین اناکاربیل) میتواند سطوح خوبی را از تسکین درد برای برخی از افراد مبتلا به نورالژی پس از هرپس و نوروپاتی محیطی دیابتی فراهم کند.
شواهد برای دیگر انواع درد نوروپاتیک
بسیار محدود است. پیامد کاهش حداقل 50% در شدت درد بهعنوان یک پیامد مفید درمان برای بیماران محسوب میشود، و دستیابی به این میزان از
تسکین درد با اثرات مفید مهمی
بر تداخل خواب، خستگی، و افسردگی، همچنین کیفیت
زندگی، عملکرد، و کار کردن همراه است.
I have been surfing on-line greater than 3 hours as of late, but I by no means discovered any attention-grabbing article like yours.
It is beautiful worth enough for me. In my opinion, if all webmasters and bloggers made good content as you did, the net will be a
lot more useful than ever before.
Does your website have a contact page? I’m having problems locating
it but, I’d like to shoot you an email. I’ve got some suggestions for
your blog you might be interested in hearing. Either way, great
blog and I look forward to seeing it expand over time.
Run the installation of the pin-up casino app download and full it.
Howdy! Do you use Twitter? I’d like to follow you if that would be okay.
I’m absolutely enjoying your blog and look forward to new posts.
Hi! I’m at work browsing your blog from my new
iphone 4! Just wanted to say I love reading through
your blog and look forward to all your posts!
Carry on the excellent work!
There is so much in this article that I would never have thought of on my own. Your content gives readers things to think about in an interesting way. Thank you for your clear information.betflik
Small business ownerss who blog about their
business enterprise can aso run into legal consequences.
diyarbakir hava durumu 15 günlük
bursa yıldırım 5 günlük hava durumu
สมัครสล็อตpg ทดลองเล่นได้แล้วที่นี่ PGTHAI.CLUB
สมัคร สล็อต pg เว็บตรง ไม่ผ่านเอเย่นต์
https://pgthai.club
สมัครสล็อตpg
I am really grateful to the holder of this site who has shared this fantastic
paragraph at here.
Good Morning everyone I want to share my talent on money making.
안전놀이터 추천 Please check this out and you will be rich
just like me!
I’m truly enjoying the design and layout of your site.
It’s a very easy on the eyes which makes it much more enjoyable for me to come here and visit more
often. Did you hire out a designer to create your theme?
Fantastic work!
сколько стоит операция по выпрямлению члена
Touche. Great arguments. Keep up the amazing effort.
Bardzo podobają mi się Twoje informacje na weblogu,
stworzyłem podobny materiał, który może
ciebie zainteresować i będę wdzięczny jeśli rzucisz swoim fachowym
okiem i dasz znać co o nim myślisz: https://denticabieleccy.pl/
Someone necessarily lend a hand to make critically
articles I’d state. That is the very first time I frequented your web
page and to this point? I amazed with the research you made to make this actual put up extraordinary.
Fantastic activity!
you are truly a good webmaster. The site loading pace is amazing.
It seems that you are doing any unique trick. Also,
The contents are masterwork. you’ve performed a fantastic process on this topic!
metrum.org (Metrology: The Forgotten Science) is the web
page devoted to typically the memory of Livio Catullo
Stecchini.
The Persian Wars, The Origin of Money inside Greece and various other historical events.
If you desire to grow your experience just keep visiting this
site and be updated with the latest information posted
here.
Get live train status, tatkal reservations for special trains, and confirmations for booking train tickets.Indian Railways’ train schedule, PNR status, live NTES running status, knowing where my
train is, and checking train status
aspirin and synthroid effects of synthroid on pregnancy over use of synthroid
We stumbled over here from a different page and thought I should check things out.
I like what I see so now i’m following you. Look forward to looking over your
web page for a second time.
PDF Suite is a very user-friendly PDF editor that can be used to view, create and edit PDFs, and has a huge
range of easy-to-use features that will help you in your day to day needs related to PDF files.
lyrica medication pregabalin max dosage lyrica for sleep
Yes, when the virtual peet craze hit I got a Tamagotchi for Christmas.
Here is my homepafe … blog
does lyrica get you high lyrica coupon card when will caremark cover pregabalin?
Great delivery. Solid arguments. Keep up the amazing
work.
metrum.org (Metrology: The Forgotten Science) is typically
the site devoted to the particular memory of Livio Catullo Stecchini.
The Persian Wars, The Origin of Money within Greece
and different historical events.
%%
%%
bactrim for parrots contraindications with bactrim bactrim f para la clamidia
Hello who is interested in hearing my story of how i became a millionaire$ 메이저사이트 Click on the
site to hear my story!
Hi, I do think your blog could be having browser compatibility problems.
When I take a look at your site in Safari, it looks fine but when opening in IE, it’s got some overlapping issues.
I just wanted to provide you with a quick heads up!
Other than that, great website!
%%
https://mengotticouture.com/shop/eyewear/women-eyewear/products/celine-edge/
Celine Edge Sunglasses
Celine Edge Sunglasses
atenolol heart rate low dosage of atenolol for migraine mode of action of amlodipine and atenolol
Great article.
synthroid and hemroids synthroid interaction with calcium need to increase synthroid
สมัครสล็อตpg ทดลองเล่นได้แล้วที่นี่
PGTHAI.CLUB
สมัคร สล็อต pg เว็บตรง ไม่ผ่านเอเย่นต์
https://pgthai.club
https://pgthai.club
https://mengotticouture.com/brands/clinique/
clinique Skin Care for women
clinique Skin Care for women
Thanks in support of sharing such a pleasant idea, article is good, thats
why i have read it completely
Hello who is interested in hearing my story of
how i became a millionaire$ 메이저사이트 Click on the site to hear my story!
Highly descriptive article, I loved that a lot.
Will there be a part 2?
First of all I want to say great blog! I had a quick question that I’d like to ask if you do not mind.
I was curious to find out how you center yourself and clear your head before writing.
I have had a tough time clearing my mind in getting my thoughts
out there. I do enjoy writing but it just seems like the first 10 to 15 minutes are usually wasted simply just trying to figure out how to begin. Any
ideas or tips? Appreciate it!
The true goal of cooking is always to nourish these
marvelous bodies we live in, to enable them to grow and express vitality and strength, to maintain them healthy and able to overcome environmental germs and bacteria.
Summarized in a word, the principle function of cooking is heath.
Hello, I enjoy reading through your post. I wanted to write a little comment to support
you.
glqbiin gbersm sljnsm ourxhb xnaomo dnrzqa
Bardzo podobają mi się Twoje informacje na blogu, stworzyłem podobny temat , który może ciebie zainteresować i będę wdzięczny jeśli rzucisz swoim fachowym
okiem i dasz znać co o nim myślisz: https://denticabieleccy.pl/
รับรองได้เลยว่าการเข้าใช้บริการที่นี่ จะสามารถทำให้ นักพนันทุกท่านติดใจกับห้องเกม กด
สล็อต ฟรี ของเรา กันได้โดยที่ คุณจะไม่อยากโยกย้ายเงินไปใช้บริการที่
อื่นได้อย่างแน่นอน เพราะว่าเว็บไซต์เดียวของเรา ที่ นั้นจบครบทุกรสชาติให้กับนักพนันทุกคนก่อนได้เลยจริงๆ และยังมีประสบการณ์ในการเล่นเกมให้กับทุกท่านได้เข้ามาใช้บริการกันได้โดยที่ ทุกคนจะต้องมีความไว้วางใจอย่างสูงที่สุด ในการเข้าใช้บริการที่ นี้แน่นอน เลยด้วย ซ้ำ และยังจะสามารถทำให้ ทุกท่านได้เข้าร่วมสนุกกับห้องเกมของเรา ทุกห้องด้วย ความมั่นใจ
This information is worth everyone’s attention. How can I find out more?
Hi there, I enjoy reading all of your post. I wanted to write a little comment to support you.
A vegan diet is a bit challenging for carnivores.
Remember, weight loss is all about diet and train. For many, meaning turning to merchandise reminiscent of train packages, trendy how-to books,
or commercialized foods and drinks, none of which are doubtless to supply protected, lasting outcomes.
Symptoms are dizziness, irritability, and nausea; the headache is a throbbing pain on the sides of the head or behind the eyes.
When it is due to wind cold, the ache is usually in the back
or the highest of the head. It accommodates two herbs specific for complications due to liver imbalances.
An effective patent remedy for the sort of headache is
Huo Xiang Zheng Qi Wan, which can also be appropriate for complications due
to inside dampness. The standard method for this pattern is Long Dan Xie Gan Wan, which
purges heat from the liver. If the underlying pattern is qi or blood deficiency,
the diet needs to be modified to incorporate more entire grains and beans, nuts, dark greens, and fresh, organic vegetables.
The traditional system for this pattern is Chuan Xiong
Cha Tiao Wan taken along with inexperienced tea.
Treatment involves soothing the liver while concurrently stimulating digestion, and an applicable remedy is Shu Gan Wan.
bursa hava durumu
The Body Shop Hyaluronic Acid Serum
I’m really enjoying the design and layout of your website.
It’s a very easy on the eyes which makes it much more pleasant for me to come here and
visit more often. Did you hire out a designer to create your theme?
Excellent work!
Although part of the appeal of betta fish is because they can be kept without the need for a large
aquarium or having a complicated filtration, betta fish care
does require some effort.
This supplement from nature accommodates an unique mix of components that reduces the event of cavities.
Really great info can be found on site. https://myspace.com/rivernation1
Howdy! Do you use Twitter? I’d like to follow you if that would be okay.
I’m definitely enjoying your blog and look forward to new posts.
Hello! Do you use Twitter? I’d like to follow you if that would be ok.
I’m undoubtedly enjoying your blog and look forward
to new updates.
bursa hava durumu
thank you so much guys i really appreciate it.
Bardzo podobają mi się Twoje informacje na blogu, stworzyłem podobny temat , który
może ciebie zainteresować i będę wdzięczny jeśli rzucisz swoim fachowym okiem i dasz
znać co o nim myślisz: https://mkbe.pl/
Whoa a lot of superb facts!
Factor clearly used!.
Hello, I enjoy reading all of your article. I like
to write a little comment to support you.
who knows what 슬롯매니악 means? If you
guess this correctly i will send you a free gift for a prize!
Etobicoke Mover #1 is a company that pursues a single goal-customer satisfaction. Our movers in Etobicoke
provide highly personalized service when moving. Our company has insurance coverage of up to $100.000!
We have specialized in local moving in Etobicoke and the district since 2001.
Also, we carry outt any mkving needs, whetther it is Long distance moving or international moving.
Our team offers you the most modern and clean trucks, the most
highly qqualified personnel and modern technologies to make your move easy and safe.small movers etobicoke
I really appreciate the information man thank you!
I always spent my half an hour to read this
blog’s articles or reviews every day along with a cup of coffee.
Handyman In Boca Raton is a local company that does all kinds of handymanservices.
We are available for all your home and business repair needs.
We offer handyman services like plumbing,roofing, electrical work, pest control, interior designing,
landscaping, and
more. We also offer emergency services and we
are available 24 hours a day.
Name:
Handyman In Boca Raton
Website:
https://handymaninbocaraton.com/
Adress:
636 NW 13th St #31, Boca Raton, FL 33486, UnitedStates
Simply wish to say your article is as amazing. The
clearness to your put up is simply spectacular and i could suppose you’re a professional on this subject.
Fine together with your permission let me to take hold of your RSS feed to keep updated with
drawing close post. Thank you 1,000,000 and please continue
the gratifying work.
May I simply just say what a comfort to find someone who actually understands what they are discussing
on the net. You actually know how to bring an issue to light and make it important.
More people must read this and understand this side of the
story. I was surprised you aren’t more popular since you certainly have the gift.
Hello NC Hello NC celebrates the people, culture, and
stories of rural Notth Carolina by waay oof neighborhood voices and wealthy media.
my page Find more info (Juliane)
It’s nearly impossible to find educated people for
this topic, however, you seem like you know what you’re talking
about! Thanks
everyone I want to share my talent on money making.
꽁머니 check this out and you will be rich just like me!
If you live blackpink from kpop please leave
me a comment in my page who your best member is because i really love lisa:)
All i wanted to know was this information. thank you and now its
my turn to give back on how to make alot of money.
Follow me ~
Let me show my appreciation through good info as well. follow for
satisfaction
Truly lots of awesome facts.
Hey! This is my first comment here so I just wanted
to give a quick shout out and tell you I truly enjoy reading your posts.
Can you suggest any other blogs/websites/forums that go over the same topics?
Thanks!
Love you all and god bless you. Thank you for this kindness
You could definitely see your skills in the work you
write. The world hopes for more passionate writers such as you
who aren’t afraid to mention how they believe. All the time go after
your heart.
Who loves BTS? i will give the first 500 a picture that
i took in korea
Nicely put, With thanks!
I’d like to find out more? I’d love to find out some additional information.
Good response in return of this query with solid arguments and describing all
about that.
Who is interested in hackers? i have many hacking services provided
Lynx DNA Laboratory is the leading DNA testing center based in Lagos, Nigeria.
Through a growing network of globally recognized partnerships that we have built
over the years, we are accredited by the United States-based GENETICA DNA Laboratories
and DNA Diagnostic Center (DDC) to deliver fast, accurate and
affordable DNA testing through our offices in major cities across
the country.
Meds prescribing information. What side effects? brand cenforce Superb what you want to recognize about medicine. Get news now.
All i wanted to know was this information. thank you and now its
my turn to give back on how to make alot of money. Follow me
~
Hello, I enjoy reading all of your post. I like to
write a little comment to support you.
Hi there! Do you use Twitter? I’d like to follow you if that
would be ok. I’m undoubtedly enjoying your
blog and look forward to new updates.
Hyella provides the best electronic medical record software for all healthcare providers in Nigeria.
Our HMIS is a suite of easily customizable electronic health record modules
that work perfectly for hospitals and clinics regardless of their specialities or
number of branches.
He was also awarded $two,022 as he won the Pennsylvania Lotttery on the net prize
drawing, aaccording to ABC27.
my homeppage Click for info; flexmon.xyz,
غسل کولو طریقه
We have several types of palm oil
عوارض روغن پالم در لبنیات
I would love to tell you a secret about putin :]
Hey! I could have sworn I’ve been to this site
before but after reading through some of the post I realized it’s
new to me. Nonetheless, I’m definitely happy I found it and I’ll be bookmarking
and checking back often!
Are There Any Side Effects To Ikaria Lean Belly Juice?
Lean belly juice, according to the makers, is proven to produce
results and has no negative side effects. We all know
that using such drinks has a disadvantage in that
weight quickly returns after treatment. Many people are turning more to these drinks as they search for quick and practical
solutions as the prevalence of obesity increases globally.
They don’t realize, however, how much patience, commitment, self-control,
and persistence are generally needed for weight loss.
People adapt to these drinks very quickly, but when they stop drinking them, they quickly revert to their pre-drink physical states.
But before taking such supplements, we advise you
to speak with your doctor or a nutritionist.
Lynx DNA Laboratory is the leading DNA testing center based in Lagos, Nigeria.
Through a growing network of globally recognized partnerships that
we have built over the years, we are accredited by the United States-based GENETICA DNA Laboratories and DNA
Diagnostic Center (DDC) to deliver fast, accurate and affordable DNA testing through
our offices in major cities across the country.
I’m really enjoying the design and layout of your website.
It’s a very easy on the eyes which makes it much more enjoyable for me to come here and visit more often. Did you hire out a developer to
create your theme? Great work!
Hello there! Do you use Twitter? I’d like to follow you if that would
be ok. I’m definitely enjoying your blog and look forward to new posts.
Multimedia Engineering
Informatics Engineering
Internet Engineering
Language Center
International Studies
Electronics Engineering
telecoms
electrical engineering
computer engineering
If you search by a city, we’ll include things like jobs inside a 25-mile radius.
my page 여성알바
지금부터 메이저사이트 관련 소개하려는데 관심
있으시면 함 보러오세요! 후회 없을겁니다 :
)
https://www.kesepian.net/
GARWA4D | LINK ALTERNATIF GARWA4D | LINK DAFTAR GARWA4D | LINK LOGIN GARWA4D | LINK WAP
GARWA4D
Digikala Indian coconut oil
ترکیب حنا و روغن بادام تلخ برای مو
Let me show you how to make some cash quickly. I mean passive income is
great right? you wont regret it man.
Application of sodium hydrosulfite
Excellent write-up. I certainly appreciate this site.
Thanks!
کاراگینان مارسل
지금부터 메이저사이트 관련 소개하려는데 관심 있으시면 함 보러오세요!
후회 없을겁니다 🙂
지금부터 메이저사이트 관련 소개하려는데 관심 있으시면 함 보러오세요!
후회 없을겁니다 🙂
Let me show my appreciation through good info as well.
follow for satisfaction
Я смотрю ежедневные новости исключительно тут
What are the 5 best Black Friday websites?
https://jclsn.medium.com/why-should-you-reinvent-yourself-6fc0d324d102?source=rss-28d8a59755af——2
Hello, I enjoy reading through your article. I wanted to write
a little comment to support you.
All i wanted to know was this information. thank you and now
its my turn to give back on how to make alot of money.
Follow me ~
I’m not sure exactly why but this site is loading incredibly slow for me.
Is anyone else having this issue or is it a issue on my end?
I’ll check back later and see if the problem still exists.
지금부터 메이저사이트 관련 소개하려는데 관심 있으시면 함 보러오세요!
후회 없을겁니다 🙂
Можно ли сдать одежду в секонд хенд за
деньги? свитер из шерсти мериноса
Fortunately, we’ve done the work — and dealt with the pop-up ads — so you
don’t have to.
Good way of describing, and good article to get information on the topic of my presentation subject matter, which i am going
to present in institution of higher education.
It’s enormous that you are getting ideas from this article as well as from our argument made at this time.
We also help our purchasers with financial institution deposit to file citizenship purposes.
สมัครสล็อตpg ทดลองเล่นได้แล้วที่นี่ PGTHAI.CLUB
สมัคร สล็อต pg เว็บตรง
ไม่ผ่านเอเย่นต์
https://pgthai.club
pgthai
All i wanted to know was this information. thank you and now its my turn to give back on how to make alot of money.
Follow me ~
I’d like to find out more? I’d like to find out some additional information.
Как сделать так чтобы свитер не кололся?
свитер 100% кашемир
Whoa all kinds of amazing material.
Appreciate it, Plenty of posts!
Hello! Do you use Twitter? I’d like to follow
you if that would be okay. I’m absolutely enjoying your blog
and look forward to new updates.
Hello there! Do you use Twitter? I’d like to follow you if that
would be ok. I’m definitely enjoying your blog and look forward
to new updates.
Hello, I enjoy reading all of your article. I like
to write a little comment to support you.
지금부터 메이저사이트 관련 소개하려는데 관심 있으시면 함 보러오세요!
후회 없을겁니다 🙂
ddc ราคาถูก,vps ราคาถูก,vps forex,ddc,รับวาง ddc,
เช่า ddc,เช่า vps,vps รายวัน,vps รายเดือน,รับวาง Dedicate Server,
เช่า Dedicate Server,เช่า Colocation,Hosting
You have made your position extremely effectively!!
In travel swiftly we create contents for
our visitors to help them select their travel destinations, by showing them the attraction tourist places, let them know very interesting information about
many countries, including historical places, tourist guides, important tips
and recommended accommodation.
In addition, we launched our youtube channel under the same name (Travel swiftly),
which includes very interesting video in 4k resolution to make our users enjoy seeing their destinations.
We hope you enjoy our content and your amazing
destination in entire world.
All i wanted to know was this information. thank you and now
its my turn to give back on how to make alot of money.
Follow me ~
All i wanted to know was this information. thank you and now its my turn to give back
on how to make alot of money. Follow me ~
Let me show my appreciation through good info as well. follow for satisfaction
royaltoto, royaltoto login, royaltoto daftar, royaltoto wap, royaltoto link, royal toto, royaltoto.com
If you love BTS and are part of the army come check the fan site!
오피사이트
I’m really enjoying the design and layout of your blog.
It’s a very easy on the eyes which makes it much more
pleasant for me to come here and visit more often. Did you hire out a developer to create your theme?
Fantastic work!
All i wanted to know was this information. thank you and now its my
turn to give back on how to make alot of money.
Follow me ~
I used to be able to find good information from
your articles.
Microgaming telah terkenal sejak tahun 90an silam dengan permainan slot, karena ketika
itu hanyalah provider ini yang memiliki game yang sangat memanjakan ini.
Lapisan masyarakat dengan umur menuju senja pastinya sangat menandai sang penyedia ini
yang telah menemani sejak dahulu kala. Game slot Slot 777 yang terkenal itu terdiri
dari nama : Mayan Princess, Gold Factory, Dragon Dance, Candy Dreams sampai Fish Party serta lainnya.
All i wanted to know was this information. thank you and now its my turn to give back on how to make alot
of money. Follow me ~
Is any body interested in making alot of money for passive income side hustle?Noone will believe me but i made soo much through 오피사이트 I would like
to share my experience 🙂
Amazing all kinds of fantastic knowledge.
Wow lots of amazing info.
All i wanted to know was this information. thank you and now its my turn to give back
on how to make alot of money. Follow me ~
와~ 진짜 내가 원하던 정보들이네. 똑같이 해줘야되겠는데, 나도
가치를 제공하고 싶은데요 그거아시나 혹시 매일 100만원 만들기 이렇게
쉽게 알수 없는 내용를 제가 나눠드리겠습니다.
한번 확인 해보시죠!
Truly a lot of very good knowledge.
I have some value that I want to return to everyone
because of all the information I was givin this whole time.
Follow me guys~
Factor well applied.!
Гарний пост, я поділився ним із друзями.
I always show love to the people writing this.
So let me give back and speak out on change your life and if you want to
truthfully see I will share info about how to become a millionaire I am always here for yall.
내가 찾고 싶은 의견들 볼수 있어서 너무 좋네요.
나도가치를 제공하고 싶은데요 그거아시나 혹시 푸틴이 우크라이나 에서 진짜 하려는 사실 이렇게 좋은 내용를 제가 가치 제공을
해드리겠습니다. 우리 서로 좋은 정보 나눠주고 이렇게 성공 합시다.
%%
Who wants to be lookin like a ballin multi millionaire?
Come chec kthis place out and cop some real looking stuff
How often can I use my nebulizer?
https://budesonideinhaler.com does symbicort contain steroids
I really appreciate your comments and reply.
I will share you a footage of russians being destroyed
in the war 🙂
Poison dope in compensation patients. Effects of Tranquillizer Abuse. https://avodart.beauty/ how much is avodart All advice here medication. Decipher information now.
Situs moontoon care terpercaya membagikan informasi terbaru
tentang dunia iGaming.
와~ 진짜 내가 원하던 정보들이네. 똑같이 해줘야되겠는데, 나도 가치를 제공하고 싶은데요 그거아시나 혹시 이쁜 여자 찾는 거 이렇게 좋은 내용를
제가 알려드리겠습니다. 한번 확인 해보시죠!
I really appreciate for this man. Can i show my inside to change
your life and if you want to findout? I will share info about how to become a millionaire I will be
the one showing values from now on.
Kudos! I value it!
Wonderful material. Thank you!
I really appreciate for this man. Can i shout out on change your life and if you
want to hear exactly I will share info about how to learn SNS marketing I will
be the one showing values from now on.
I always show love to the people writing this. So let me give back
and tell you my secret ways on change your life and if you want to
findout? I will share info about how to change your life I am always
here for yall.
I have some value that I want to return to
everyone because of all the information I was givin this whole time.
Follow me guys~
Situs bocoran slot
adalah situs betting terbaik di Indonesia 2022-2023 dengan ratusan game slot terbaru dan rtp
tinggi. Nikmati bonus menguntungkan dan jaminan anti rungkat.
Janji Gacor!
Regards. Ample content!
Who loves to make a fortune just like me? If you are
interested come hear me out yall!오피
Excellent goods from you, man. I’ve understand your stuff previous to and you are just extremely great.
I really like what you’ve acquired here, really like what you are saying and the way in which you say it.
You make it entertaining and you still take care of to keep it sensible.
I cant wait to read much more from you. This is actually a terrific website.
澳娱有线
Situs tangan judi terpercaya membagikan informasi terbaru tentang dunia iGaming.
Hello! This is my first comment here so I just wanted to give a quick shout out and say I genuinely enjoy
reading through your articles. Can you recommend any other blogs/websites/forums that deal
with the same subjects? Thank you so much!
I always show love to the people writing this. So let me give back and give out my secrets to change your life and if you want to hear exactly I will share
info about how to become a millionaire I am always here
for yall.
在线赌球
Hello! Do you use Twitter? I’d like to follow you if
that would be ok. I’m undoubtedly enjoying your blog and look forward to new posts.
Nicely put, With thanks!
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
https://vk.com/club216571129
https://vk.com/club217205332
What are the best medicines for asthma symbicort mg?
Bardzo podobają mi się Twoje informacje na weblogu, stworzyłem podobny temat , który
może ciebie zainteresować i będę wdzięczny jeśli rzucisz swoim fachowym okiem i dasz znać co o nim
myślisz: https://rafalszr.ssd-linuxpl.com/
Hi there! Do you use Twitter? I’d like to follow you if that would be okay.
I’m definitely enjoying your blog and look forward to new posts.
I believe people who wrote this needs true loving because it’s a blessing.
So let me give back and with heart reach out change your life and if you want to have a
checkout I will share info about how to become a millionaire Don’t forget..
I am always here for yall. Bless yall!
I really appreciate your comments and reply. I will share you a
footage of russians being destroyed in the war 🙂
와~ 이건 진짜 대박이네요. 제가 그토록 원하던 정보들이네.
저또한 똑같이 해줘야되겠는데, 저도 알려드리고 싶은데요
그거아시나 혹시 쉽게 돈 만들기 말도 안되는 이야기라고는 하지만 이렇게 좋은 내용를 저만 알고 있는 방법이 있는데 제가 알려드리겠습니다.
한번 믿어보시고 확인 해보시죠!
What is a better way to give a thumbs up to you? Who else these days gives awesome commentary
like this. I really appreciate it man!
Из-за непродуманного старта компания лишилась многих клиентов.
%%
Lovely stuff, With thanks.
Very well voiced truly. .
https://vavada.bitbucket.io/
Yall already know how much iwant to give a subscribe or a follow for
this. Let me give love to really amazing stuff and if
you want to know whats up? I will share info about how to make a
fortune check and follow me bros!
We also help our purchasers with financial institution deposit
to file citizenship purposes.
Но истинное лицо он показывает только
в шлейфе, когда раскрываются ветивер, амбра и табачные листья.
I’m not sure why but this site is loading incredibly slow for me.
Is anyone else having this issue or is it a problem on my
end? I’ll check back later and see if the problem still exists.
Great article. I’m facing some of these issues as well..
Putin just announced he will stopthe war. Do you know why?
Let me tell you whats going on easy understanding!
Good article. I certainly appreciate this site.
Keep it up!
Hi! Do you use Twitter? I’d like to follow you if that would be ok.
I’m definitely enjoying your blog and look forward to new updates.
Having read this I thought it was rather informative.
I appreciate you taking the time and effort to put this short article
together. I once again find myself spending a lot of time both reading and
commenting. But so what, it was still worth it!
Hey! Do you use Twitter? I’d like to follow you if that
would be ok. I’m undoubtedly enjoying your blog and look forward to new posts.
와~ 이건 진짜 대박이네요. 제가 그토록 원하던
정보들이네. 저또한 똑같이 해줘야되겠는데, 저도 보답해주고 싶은데요
그거아시나 혹시 푸틴이 우크라이나 에서 진짜 하려는 사실 말도 안되는 이야기라고는 하지만 이렇게 멋진 내용를 저만 알고 있는
방법이 있는데 제가 드리겠습니다.
한번 믿어보시고 확인 해보시죠!
Awesome forum posts, Kudos.
You actually expressed this fantastically.
https://arenda-shatrov-ty1.ru
NHL has been a popular sport in various parts of the world for decades.
It is now becoming a huge bet for today’s sporting events
https://www.yteamsshop.com/.
NHL feature distinctive colors and logo design. For the
latest in sports apparel.
https://arenda-shatrov-ty1.ru/arenda-shatrov-tentov
와 너무 멋져요! 제가 찾고 있던
정보가 있었는데, 정확하고 상세한 정보를 제공해주셔서 감사합니다
Krytykuj przyjaciela w cztery oczy, a chwal
zawsze przy swiadkach i podwladnych. Leonardo da Vinci & Tm.
Разрывная прочность всего жгута системы СИП-4 достаточно велика, однако
для прокладки ВЛ данный провод
не подходит.
malaria treatment wiki plaquenil adverse effects malaria pills over the counter
You actually mentioned that superbly.
Whoa all kinds of terrific data!
Yall already know how much iwant to give a subscribe or a follow for this.
Let me show my secrets on really amazing stuff and if you want to get a
peek? I will share info about how to learn SNS marketing check and follow
me bros!
Bahis Siteleri Güvenilir mi? Bahsi geçen güven ihlali aslında kolay kolay onarılmayacak bir tablodur.
Ancak, bahis siteleri konusunda dikkatli olursak ve
uygun incelemeleri yaparsak, sorun yaşamayız.
Ve ilk etapta daha düşük bütçe ile yatırım yapmak üzerine bahse girersek, bu bizi olası
büyük memnuniyetsizliklerden koruyacaktır.
Siteye yaklaşırken hangi köşelerin işaretlenmesi gerektiğini yukarıda belirtmiştik.
O yüzden bunu tekrarlamanın bir anlamı yok. Ancak,
genel olarak gerici piyasa tepkisine aldanmayın. Hangi kullanıcı incelemeleri çok kötü bir şekilde manipüle
edilebilir. Çünkü birçok site ücretli inceleme satın alıyor.
Bu aynı zamanda web siteleri tarafından olumsuz yorum yazdırma teknikleri şeklinde de kullanılır.
Deneme Bonusu veren bahis siteleri, güvenilirlik aşamasını geçtikten sonra daha da önem
kazanır. Ne dedik? İçerik tek başına yeterli değildir.
Ancak güvenilirlik yeterli değildir. İkisini birleştirmek,
bir araya getirmek, barışmak lazım. Bu her site için geçerli değildir.
Bu başarıyı frenlemek önemlidir. Amacımız, bu iki fenomeni
bir araya getirme yeteneğine sahip olanları çekmek.
https://telstarnet.com/
I went here and I’ve been to many different
treatment centers but white sands were the best decision I had made in a very long time.
I’m extremely grateful for them and the opportunities that
have opened up for me. I would recommend them to anyone.
Amazing info, Appreciate it.
I love when you talk about this type of stuff in your blog.
Perhaps could you continue this?
Regards. I value this.
Good article. I certainly love this site. Keep it up!
I want to share a crazy life changing site that you will not ever regret.
Come at me to find out
I positively like this record, i did not appreciate a kismet
of the things that you posted in here. i ahve much more new news regarding these topics and topics related to it.
some people may find it implacable to understadn the english language but i find it rather
calm an eye to the confidentiality that has discover to be what is todays policy.
Thanks a lot! A good amount of facts.
Kudos. Plenty of information.
Escaping the matrix needs a cheat code on making cash quickly.
Anyone interested? I will share this once and only once
오피사이트 This
site is secure and very fun with many games.
https://propiska-msk03.ru
Hi there, I enjoy reading all of your article. I wanted to write a little comment to support you.
I believe people who wrote this needs true loving because it’s a blessing.
So let me give back and show you exactly how to do change your
life and if you want to with no joke truthfully see I will share
info about how to find good hackers for good price Don’t forget..
I am always here for yall. Bless yall!
Bardzo podobają mi się Twoje informacje na blogu, stworzyłem podobny temat , który może ciebie zainteresować i
będę wdzięczny jeśli rzucisz swoim fachowym okiem i dasz znać co o
nim myślisz: https://grupaone.pl/
https://propiska-msk03.ru
Yall already know how much iwant to give a subscribe or a follow for this.
Let me show my secrets on really amazing stuff and if you want to get a peek?
I will share info about how to change your life check and
follow me bros!
You revealed that effectively.
You know what? This is some cool stuff that ive just noticed.
Keep it up man ~
You said this fantastically!
You stated that perfectly.
I’m really enjoying the design and layout of your blog.
It’s a very easy on the eyes which makes it much more enjoyable for me
to come here and visit more often. Did you hire out a
designer to create your theme? Excellent work!
Yall already know how much iwant to give a subscribe or
a follow for this. Let me tell you my ways on really amazing stuff and
if you want to know whats up? I will share info about how to make passive income check and follow me bros!
Putin just announced he will stopthe war. Do you know why?
Let me tell you whats going on easy understanding!
Nicely put, Thanks a lot!
Fine content Thanks a lot.
와 너무 멋져요! 제가 찾고 있던 정보가 있었는데, 정확하고 상세한 정보를 제공해주셔서 감사합니다
Howdy! Do you use Twitter? I’d like to follow you
if that would be ok. I’m definitely enjoying your blog and look forward to new posts.
People say money isn’t happiness and I agree with
it. But the fact is Who loves to make a fortune just
like me? If you are interested come hear me out yall!오피
Appreciate it, Ample postings.
You explained this fantastically!
You explained this superbly.
You actually revealed this terrifically!
What is a better way to give a thumbs up to you?
Who else these days gives awesome commentary like this. I really appreciate it man!
Nicely put. Kudos!
Valuable knowledge With thanks!
Thanks in support of sharing such a nice thinking, paragraph is good,
thats why i have read it entirely
I have a lot of ways toto find the amzing love of my life
online so please come check me out to get awesome value also
biology exploring the diversity of life 5th edition pdf reddit
와~ 이건 진짜 대박이네요. 제가 그토록 원하던 정보들이네.
저또한 똑같이 해줘야되겠는데, 저도 알려드리고 싶은데요 그거아시나 혹시 이쁜 여자 찾는 거 말도 안되는 이야기라고는 하지만 이렇게 쉽게 알수 없는 내용를 저만 알고 있는 방법이 있는데 제가 드리겠습니다.
한번 믿어보시고 확인 해보시죠!
Amazing information With thanks!
Hello there! This is my first comment here so I just wanted to give a quick shout out and say I
genuinely enjoy reading your posts. Can you recommend any other blogs/websites/forums that cover the same topics?
Thanks!
Many thanks, An abundance of content!
Putin just announced he will stopthe war. Do you know why?
Let me tell you whats going on easy understanding!
Awesome info, Many thanks!
Wow all kinds of amazing info!
Jasper is the means to go if you’re looking for a means to generate high-grade material quickly.
The AI innovation is amazing, as well as the outcomes promote themselves.
I have a lot of ways tofind a way to hack forgotten instagram so please
come check me out to get awesome value also
Play Solitaire online, 100% free. Full screen with no download
or registration required. 500+ games.
Yall already know how much iwant to give a subscribe or a follow for this.
Let me tell you really amazing stuff and if you want to know whats
up? I will share info about how to make passive income check and follow me bros!
online sabong log in
sabong live login
sabong international log in
e-sabong login
talpak online sabong
wpc15 sabong log in
Nicely put, Regards.
Can you tell us more about this? I’d love to find out some additional information.
Hi mates, good article and good urging commented
here, I am in fact enjoying by these.
I simply wanted to appreciate you once again. I am not sure the things I would have tried in the absence of these tricks shown by you regarding such a industry. Completely was a very hard situation for me, however , being able to see the very professional fashion you treated it took me to weep with gladness. I will be happier for the assistance and even expect you know what an amazing job that you’re carrying out educating others using your website. I know that you have never come across all of us.
Great delivery. Solid arguments. Keep up the
amazing spirit.
Very good article. I am facing some of these issjes as well.. https://storage.googleapis.com
Hi! Do you use Twitter? I’d like to follow you if that would be okay.
I’m absolutely enjoying your blog and look forward to
new updates.
I am glad to be a visitor of this stark web site, regards for this rare information!
Hi there! Do you use Twitter? I’d like to follow you if that would be okay.
I’m undoubtedly enjoying your blog and look forward to new
updates.
Highly informative, look ahead to coming back.
https://www.swiebodzin.tv
%%
Point nicely considered..
Nicely put. Thank you.
Tips nicely used!!
Hello, I enjoy reading through your post.
I wanted to write a little comment to support you.
SNS 계정 복구,인스타 해킹 복구, 페이스북
해킹 복구, 구글 계정 복구, 네이버
계정 복구, 카카오톡 해킹 복구,인터넷, 모바일, PC 각종 해킹 피해
관련된 상담 서비스,바이러스 검수 서비스,악성 링크 유무 및 검사 서비스,디지털 포렌식,데이터
손상 및 복구 서비스,해킹 점검, 네트워크 검수 서비스, 네트워크 관제, 패킷 분석, 공유기 해킹, 기술지원으로 원격 접속 후
안전성 검사 서비스. 해킹 종합 솔루션 참새왕 해킹 피해 상담 서비스
네이버 지식인 상담 및 올바른 답변을 제공하는 것을 시작으로 현재 지식인 해킹 전문가 랭킹 3위로 유지하고
있습니다. 2022년 3월 처음 지식인 엑스퍼트 통해 상담 서비스를 제공해드리기 시작했고 어린 학생
부터 직장인, 인플루언서, 컴퓨터가 어려운 어르신까지 포괄적인 범위로 상담을
진행 해오면서 의뢰인분들께서 겪고 있는 모든 고충들과 상황들을 해결하였습니다.
Awesome content Many thanks.
Nicely put, Regards.
BBB is the 1 and unique dark and arab gathering
found in paris since 16 ages.
Every sunday at folies pigalle’s tavern from 10 pm till morning the special and incredible celebration with beautifull dark-colored guys and arabe rnb songs and arabic mixes, remove present and diva shows.
Truly quite a lot of terrific material!
Some research from 2011 indicated that consuming probiotic yogurt could improve cholesterol levels
in folks with type 2 diabetes. While pointing to an absence of scientific testing of the procedure’s purported
advantages, the medical establishment has additionally uncared for to provide a lot in the way of analysis on this system.
However, analysis suggests that low carb diets may be helpful.
This may increasingly help folks with diabetes enhance glycemic control and maintain a average weight.
It might also assist folks with diabetes maintain a reasonable
weight. Where a food falls on the GI suggests the effect that food
might have on blood sugar ranges, but the GI
does not take all points of the food, including portion dimension, under consideration. A
low carb eating regimen can assist cut back
cravings, decrease blood sugar levels, and increase
vitality. Unhealthful fats, equivalent to saturated and trans fats,
can make an individual with diabetes really feel
worse. This may occasionally contain fastidiously accounting for carbohydrates to
guantee that the person has enough power however retains their blood sugar ranges beneath management.
Any such glaucoma may not trigger any signs till it’s further along and you’ve got major imaginative and prescient loss.
You have many signs of diabetic ketoacidosis. These two symptoms go
hand in hand and are some of “your physique’s ways of attempting to manage high blood sugar,” mentioned
Maria Collazo-Clavell, MD, Professor of Medicine within the Department Internal Medicine at the Mayo Clinic.
Thanks a lot. Useful information!
I’m truly enjoying the design and layout of your website.
It’s a very easy on the eyes which makes it much more enjoyable for me to come here and visit more often. Did you
hire out a developer to create your theme? Excellent work!
Временная регистрация в Москве
костюм для квадроцикла
This is nicely put. !
have good day
find best similar song net by track
African American, Alaska Native, American Indian, Asian American, Hispanic/Latino, Native Hawaiian, or Pacific Islander.
Diabetes is extra frequent in people who find themselves African American, Hispanic/Latino, American Indian, Asian American, or Pacific Islander.
7 Who’re the people at risk for prediabetes?
Read on to seek out out about the risk components, and modifications you may make to scale back your threat.
Anyone experiencing fluctuations in glucose levels and excessive blood sugar within the morning ought
to discuss this with a doctor before making any adjustments
to their insulin treatment. Projections present that the growing
number of people with diabetes could result in practically 39,000 additional individuals suffering a coronary heart attack in 2035, over 50,000 experiencing a stroke, while
one in six hospital beds now occupied with somebody with
diabetes. An A1C quantity of six. The greater your glucose
levels have already been in the last couple of months, the
greater your A1C stage can find yourself being.
Hello! This is my first comment here so I just wanted to give a quick shout out and tell you I really enjoy reading through your posts.
Can you suggest any other blogs/websites/forums that cover the same
subjects? Appreciate it!
Just wish to say your article is as surprising.
The clearness in your post is just excellent and that i can assume you
are an expert in this subject. Well with your permission let me to take
hold of your feed to keep updated with approaching post.
Thank you a million and please carry on the enjoyable work.
Wonderful material, Thanks!
You know what? This is some cool stuff that ive just noticed.
Keep it up man ~
Cheers, Quite a lot of posts!
I believe people who wrote this needs true loving
because it’s a blessing. So let me give back and show my hidden information on change your life and if
you want to with no joke truthfully see I will share info about how to make passive
income Don’t forget.. I am always here for yall.
Bless yall!
You have made your point extremely clearly.!
Useful forum posts Regards!
I want to share a crazy life changing site that you will not ever regret.
Come at me to find out
I want to share a crazy life changing site that you will not
ever regret. Come at me to find out
Regards! An abundance of postings.
I believe people who wrote this needs true loving because it’s
a blessing. So let me give back and tell you exactly how to do change your
life and if you want to have a checkout I will share info about how to
make a fortune Don’t forget.. I am always here for yall.
Bless yall!
Regards! I enjoy it!
Es importante hablar con su médico sobre su riesgo individual de
desarrollar cáncer de mama y qué medidas puede tomar para
prevenirlo.
I believe people who wrote this needs true loving because it’s a
blessing. So let me give back and give out my secrets to change your life and if you want to seriously get to hear I will share info about how to change your life Don’t forget..
I am always here for yall. Bless yall!
Truly all kinds of valuable advice!
I’m truly enjoying the design and layout of your blog.
It’s a very easy on the eyes which makes it much more
pleasant for me to come here and visit more often. Did you hire out a
designer to create your theme? Outstanding work!
I believe people who wrote this needs true loving because it’s a blessing.
So let me give back and give my value on change your life and if you want to with no joke truthfully see I
will share info about how to become a millionaire Don’t forget..
I am always here for yall. Bless yall!
Wow that was odd. I just wrote an very long comment but after I clicked submit
my comment didn’t show up. Grrrr… well I’m not writing all that over again. Anyway, just wanted to say great blog!
Lovely content. Kudos!
Incredible a good deal of very good facts!
I believe people who wrote this needs true loving because it’s a blessing.
So let me give back and tell you exactly how to do change your life and if you want to with no joke truthfully see
I will share info about how to find good hackers for good price Don’t forget..
I am always here for yall. Bless yall!
Regards, Terrific information.
You stated it very well!
Wow a good deal of superb material!
Appreciate the recommendation. Let me try it out.
You actually explained that terrifically.
You’ve gotten good information in this article.
www
Nicely put, Thanks a lot!
You actually explained this terrifically!
you’re in reality a good webmaster. The web site loading speed
is incredible. It kind of feels that you’re doing any unique
trick. Also, The contents are masterwork. you have performed a magnificent activity
in this matter!
Binsion Nclex RN offers traning to nclex candidates/inter national nurses/nursing students to easily pass the nclex exam on theri first try.Binsion has timed mutiple types of test modes and practice modes with image based explanations and computer adaptive test (CAT) facility.
You will gain high knowldge and the AI powered app will help you improve your critical thinking skills to
answer any kind of questions once you finished.
60% discount is offered by the company which cost you only price of
a cup of tea.
https://binsion.com – BINSION
Bonus buy slots are online slots where you can pay a premium in exchange for instant access to their main bonus feature; usually the
free spins round. Also known as “feature buy”
You know what? This is some cool stuff that ive just noticed.
Keep it up man ~
มหาดไทย สืบสานพระปณิธาน ‘พระองค์หญิง’ อนุรักษ์ภูมิปัญญาและพัฒนาต่อยอดผ้าไทยสู่ความยั่งยื…
I believe people who wrote this needs true loving because it’s a blessing.
So let me give back and show my appreciatation change your life and if
you want to have a checkout I will share info about
how to make passive income Don’t forget.. I am always here for
yall. Bless yall!
At this moment I am going to do my breakfast, once having my breakfast
coming over again to read additional news.
I want to share a crazy life changing site that you will not ever regret.
Come at me to find out
I’d like to find out more? I’d want to find out more details.
What is a better way to give a thumbs up to you? Who else these days gives awesome commentary like
this. I really appreciate it man!
You’ve made your point extremely nicely!.
Thank you for your whole hard work on this site.
It has been an inspiration for me. I’ve passed this on to a buddy of mine.
Wow all kinds of terrific advice.
Excellent write-up. I certainly love this site. Continue the good work!
If you are going for best contents like I do, just pay a visit
this site all the time since it offers feature contents, thanks
Party room
It’s impressive that you are getting ideas from this article
as well as from our argument made at this time.
Regards, Lots of postings!
E-cigarettes
Parimatch oferuje
wiele sposobów wpłacania i wypłacania zaliczek.
I’m now not sure where you are getting your info, but great topic.
I must spend some time studying more or figuring out more.
Thanks for excellent information I was on the lookout for this information for
my mission.
Superb material. Appreciate it.
Factor clearly applied!.
If you wish for to grow your familiarity just keep visiting this
site and be updated with the most recent news posted here.
pathophysiology of heart disease an introduction to cardiovascular medicine pdf
Kasyno Slottica ma licencję Curacao, która upoważnia kasyno do prowadzenia działalności.
I’m truly enjoying the design and layout of your website.
It’s a very easy on the eyes which makes it much more enjoyable for me to
come here and visit more often. Did you hire out a developer to
create your theme? Outstanding work!
Regards, An abundance of info!
Party room
I believe people who wrote this needs true loving because it’s a blessing.
So let me give back and tell you exactly how to do change your life and if you want to with no joke truthfully see I will share info about how to make a
fortune Don’t forget.. I am always here for yall. Bless yall!
Hi there! Do you use Twitter? I’d like to follow you
if that would be ok. I’m definitely enjoying your blog
and look forward to new posts.
I believe people who wrote this needs true loving because it’s
a blessing. So let me give back and show back my secrets on change
your life and if you want to have a checkout I will share
info about how to make a fortune Don’t forget.. I am always here for yall.
Bless yall!
เราได้แนะนำเครื่องฟอกอากาศที่ดีที่สุดในท้องตลาดเพื่อช่วยลดอันตรายจากการปนเปื้อนในอากาศในบ้านของคุณ PHILIPS
รุ่น AC1215, Xiaomi Mi Air Purifier 3C, Tefal รุ่น PT3030FO, Philips รุ่น AC0820,
Blueair HealthProtect™ รุ่น 7770i, Philip
Vitashield AC2887 และ Air filter 3M Filtrete Ultra Slim FAP04 เป็นเพียงบางส่วนของเครื่องฟอกอากาศยอดนิยมที่เราแนะนำ .
เครื่องฟอกอากาศแบบพกพายังเป็นทางเลือกที่ยอดเยี่ยมสำหรับผู้ที่ต้องการให้อากาศบริสุทธิ์ติดตัวไปทุกที่
การสำรวจลดความเสี่ยงของอันตรายจากการปนเปื้อนในอากาศที่จัดทำโดยสำนักงานคุ้มครองสิ่งแวดล้อมของสหรัฐฯ ระบุว่าอุปกรณ์ประเภทนี้มีประโยชน์อย่างเหลือเชื่อสำหรับการปรับปรุงคุณภาพอากาศภายในอาคาร
Excellent items from you, man. I have take into account your stuff previous to
and you’re just too excellent. I really like what you’ve bought right here, really like what you’re stating
and the way by which you say it. You make it enjoyable and you still
care for to keep it wise. I cant wait to read far more from you.
This is actually a great website.
Istnieją różne metody, które pozwolą ci wpłacać i wypłacać gotówkę z
Parimatch Com.
A fairly bouquet made up of bright pink germini, pale yellow chrysanthemum, blue
iris and delicate white alstroemeria.
They looked gorgeous on arrival and stayed at their finest for
almost every week.
Alcune delle varianti del blackjack disponibili su LeoVegas.
it supportano fino a tre mani alla volta.
There are $400 and $500 phones with flashier standout features than the Google Pixel 5A.
The Samsung Galaxy A52 5G features a giant 6.5-inch screen with 120Hz refresh rate.
Wow. Now I’m wondering how many individuals would use a
phone on the market? Google reviews are subsequently
ripe for defamation claims, with damaging reviews usually viewed by multiple people online, which may have a substantial impact on the
status of the reviewee. Fake critiques are a rising downside for
these of us that own small companies. Are there defences to defamation? I believe you’ve got simply
shot the recognition of GMB posts (what there is) in the foot!
There are a couple of drawbacks to notice: its security assist coverage is good but not class-main. It may also be defamatory if there is a
reference to a class of individuals. 2. Sharing files:
Google Drive permits customers to simply share recordsdata with others, both by sending a hyperlink or
by inviting particular folks to view or edit the file.
Helpful knowledge Kudos!
Very descriptive article, I liked that bit. Will there be a part 2?
Terrific material, Thanks a lot.
Kudos, Plenty of knowledge!
bicikli bolt kdnp Eladóink évtizedes tapasztalata, tudása és termékismerete segítségével biztosan megtalálod a számodra legjobban megfelelő biciklit, cipőt, ruházatot,
kerékpáros kiegészítőket, szerszámokat, alkatrészket.
Folyamatosan arra törekszünk, hogy a szolgáltatásaink maradéktalanul a legjobb minőséget képviseljék.
Kerékpár üzletünkben található raktárkészlet nem egyezik meg a
webáruház kínálatával.
Warto zauważyć, że w przypadku wycofania dużej kwoty czas przetwarzania wniosku w
kasynie slottica casino może wynieść 73 godziny.
Thank you. I appreciate this!
My family members every time say that I am wasting my time here at net, except I know I am getting
knowledge everyday by reading such nice content.
Just want to say your article is as astonishing.
The clearness for your put up is simply great and that i could think you’re an expert on this subject.
Well along with your permission let me to seize your feed to
stay updated with drawing close post. Thanks a million and
please keep up the gratifying work.
Terrific content, Cheers.
BoostMyInsta Instagram
str.Diego 13, London,
E11 17B
(570) 810-1080
tiktok comments
You actually stated it exceptionally well.
Superb info, Kudos!
Passing on knowledge helps to create a more educated society.
I believe that by giving back, I am creating
a more equitable and just society.
Indeed, study suggests that women can expertise backlash
when they negotiate also aggressively.
my page 요정알바
Your method of explaining all in this post is really nice, all be able to simply know it, Thanks
a lot.
Good article. I’m facing some of these issues as well..
Incredible lots of great material.
What is the main cause of inflammation in the body plaquenil and alcohol
Having read this I believed it was rather informative.
I appreciate you taking the time and effort to put this
article together. I once again find myself
spending a significant amount of time both reading and commenting.
But so what, it was still worth it!
Appreciate the recommendation. Let me try it out.
Helpful material Thanks a lot.
Русское домашнее частное видео
Asking questions are in fact nice thing if you are not understanding
anything totally, except this post offers good understanding yet.
Hello! Do you use Twitter? I’d like to follow you if that would be
ok. I’m undoubtedly enjoying your blog and look forward to new
updates.
I’d like to find out more? I’d love to find out more
details.
There is certainly a lot to learn about this issue.
I love all of the points you’ve made.
Migrants in Texas trailer tragedy died seeking better lives Mariners should
alter plans to avoid these hazardous conditions. Remain in port,
seek safe harbor, alter course, and/or secure the vessel for severe conditions.
Part 203(b)(2)(A) makes immigrant visas obtainable to such people provided that their “services within the sciences, arts, professions or business are sought by an employer in the United States.
Can you tell us more about this? I’d love to find out
more details.
slot gacor
Superb facts. Many thanks!
Reliable facts, Thanks a lot.
Wow lots of excellent information!
Pills information leaflet. Short-Term Effects. walmart store pharmacy locator To the fullest extent intelligence about drugs. Injure here.
Very good info. With thanks.
Русское домашнее частное видео
Nicely put, Kudos!
Wonderful info Appreciate it.
Reliable data Kudos.
wonka oil pen
Kudos. Fantastic stuff!
if i were a writer essay writing an opinion essay how to write an about me section for a website
Good knowledge Regards.
Amazing a lot of fantastic information.
Cheers. I enjoy this!
how to write an about me page for a blog essay writer for you did john legend write all of me for his wife
Hi! I’m at work surfing around your blog from my new iphone 4!
Just wanted to say I love reading through your
blog and look forward to all your posts! Keep up the great work!
You definitely made the point.
Hey! Do you use Twitter? I’d like to follow you if that would be okay.
I’m absolutely enjoying your blog and look forward to new
posts.
Thanks a lot. Loads of facts.
{
Nicely put. Cheers!
writes essays for you write my argumentative essay for me hire writer for essay
Veteran’s preference indicates if two applicants are
finalists for a position, and equal in all respects, the veteran would be offered the job.
La educación sobre la prevención del cáncer de mama es fundamental para garantizar un futuro más saludable para todas
y cada una de las mujeres.
Good article. I am facing a few of these issues as well..
Can you tell us more about this? I’d like to find out more details.
Enjoyable post I tend to be of the same mind with most of what you wrote.
I would love to see more posts on this. I will bookmark and come back.
I believe people who wrote this needs true loving because it’s a blessing.
So let me give back and show you exactly how
to do change your life and if you want to with no joke truthfully
see I will share info about how to change your life Don’t forget..
I am always here for yall. Bless yall!
With thanks, Loads of information!
Great blog! Do you have any hints for aspiring writers?
I’m planning to start my own site soon but I’m
a little lost on everything. Would you advise starting
with a free platform like WordPress or go for a paid option?
There are so many options out there that I’m totally confused ..
Any tips? Thanks a lot!
Great delivery. Solid arguments. Keep up the great spirit.
Good day! Do you use Twitter? I’d like to follow you if that would be ok.
I’m definitely enjoying your blog and look forward to new posts.
Your method of explaining everything in this post is
actually nice, all can without difficulty understand it, Thanks a lot.
Great article. I’m going through a few of these issues as well..
You have made the point!
how much are muha med carts
willy wonka cart
Howdy would you mind letting me know which webhost you’re working with?
I’ve loaded your blog in 3 different browsers and I must say this blog
loads a lot quicker then most. Can you suggest a good internet hosting provider at a honest price?
Many thanks, I appreciate it!
Personally Im impressed by the quality of this.
Generally when I come across these sort of things I like to post them on Digg.
This article probably wont do well with that crowd.
Ill take a look around your site though and submit something else.
I believe people who wrote this needs true loving because it’s a blessing.
So let me give back and show my appreciatation change your life and if you want to seriously get to
hear I will share info about how to learn SNS marketing Don’t
forget.. I am always here for yall. Bless yall!
Про психов фильмы
Good article. I will be going through a few of these issues as well..
Hello, I enjoy reading through your article. I like to write a
little comment to support you.
Я ебу
slot gacor, situs slot gacor, slot gacor hari ini, slot gacor 2023, link slot gacor, bocoran slot gacor, rtp slot gacor,
gacor slot, slot gacor malam ini, slot gacor gampang menang, slot gacor maxwin, slot
online gacor, daftar slot gacor, slot gacor pragmatic, slot gacor terbaru,
info slot gacor, bocoran slot gacor hari ini, info slot gacor hari ini, https://sister.unimor.ac.id/slot-pulsa/
Thanks a lot! Plenty of forum posts!
Many thanks, I enjoy this!
Lovely content. Kudos.
There’s certainly a lot to know about this issue. I really
like all of the points you have made.
Hello, I enjoy reading through your post. I like to write
a little comment to support you.
Valuable content Cheers.
Darknet hackers and surface web hackers often have very distinct client bases that don’t often overlap, so the type of service
you’re looking for will heavily dictate where you need to find your
hacker.
Thank you. I value this.
I’m truly enjoying the design and layout of your blog. It’s a very easy on the eyes which makes it much more enjoyable for me to come here and visit more often. Did you hire out a developer to create your theme?
Excellent work!
Hey there! Do you use Twitter? I’d like to follow you if that would be ok.
I’m undoubtedly enjoying your blog and look forward to
new updates.
This site really has all of the information and facts I needed about this subject and didn’t know who to
ask.
Welcome to Minimal Mars Company! We are dedicated to providing our customers
with the latest and most innovative products to bring comfort to your doorstep.
Our online store offers unique finds that will speed up your
daily routine and ignite your creativity. Our mission is to offer affordable and
cutting-edge items that cater to your needs.
At Minimal Mars, we value your input and our customers
are the driving force behind our product selection. We want to hear your thoughts and feedback so that
we can continue to improve our products and services. Your
satisfaction is our top priority.
We have a wide range of products that cater to your every
need. For pet lovers, we have the 2 in 1 Portable Pet Toilet Picker, the Cat Brush Gloves,
the Cat Wand Toy, the Dog Supplies Pet Cleaner Portable, and the Pet Car
Seat Belt, just to name a few. These items will make taking care of your
furry friends so much easier.
We also have products for tech enthusiasts.
Our 3 in 1 Grip Anti-shake Multifunctional Phone Holder, Auto LED USB Magnetic Wood Wireless Night Light
Corridors Porch Lights, GPS Dog Tracker, and Quicksand Astronaut Bracket Phone Case for iPhone are just a
few of our popular tech products.
For those who love to cook, we have the Adjustable Silicone Clip On Strainer for Pots, Pans, and Bowls – Kitch,
Digital Food thermometer, Egg Timer Kitchen Gadget Color Changing
Boil Eggs Thermometer, Lemon Squeezer, and Spiralizer Vegetable Slicer – Onion Chopper
with Container – Pro Food. These items will make meal prep a
breeze.
Our Home & Garden products include the Fireworks Bubble Machine, Grow Lights for
Indoor Plants-3 Heads, Heavy Duty Furniture Lifter 4 Appliance Roller Sliders
with 660 lbs Load Capacity, and Refrigerator Organizer Bins
Soda Can Dispenser. These items will make your home more
comfortable and organized.
We also have products for kids and toys such as the Baby Elephant Cuddle Pillow, Kids Non Slip Crawling Elbow Infants Toddlers Baby
Accessories Smile K, and LCD Writing Tablet for Kids.
At Minimal Mars, we offer a Cancellation Policy and
a Shipping Policy that is customer-friendly. We also have a Track Order Status page where
you can check the status of your order at any time.
So come and join us today and experience the magic of
Minimal Mars Company. We are committed to bringing you the best products and services possible.
Your satisfaction is our guarantee.
It’s impressive that you are getting ideas from this article as well as from our dialogue
made at this time.
Hello, I enjoy reading all of your article. I wanted to write a little comment
to support you.
Hey there! Do you use Twitter? I’d like to follow you if that would be okay.
I’m definitely enjoying your blog and look forward to new updates.
Great article. I will be going through some of these issues
as well..
If you are going for best contents like me, only visit this
web site every day since it presents feature contents, thanks
The facility of the loan is perfect when you need money immediately for coping with unavoidable needs.
A better way to take out a personal loan is to
apply with several lenders so that you can have a choice to make a good decision. These loans offer a
bigger loan amount in the range of.
I’d like to find out more? I’d love to find
out some additional information.
порнуха в hd
Wow lots of valuable material.
slot5000, slot 5000, situs slot 5000, slot5000 alternatif link
baru, slot 5000 tanpa potongan, slot gacor, slot online, slot gacor terpercaya,
slot gacor hari ini, slot gacor malam ini, situs slot
gacor, https://sister.polibatam.ac.id/slot-5000/
Can you tell us more about this? I’d like to find out more
details.
Sadisflix et le seul est l’unique Site de Streaming 100% Français et Gratuit qui propose une grande variété de Films et de Séries
en qualité HD. Découvrez notre catalogue exclusif et profitez
d’un divertissement illimité. Nous vous offrons une sélection de contenus adaptée à vos préférences et à vos envies, ainsi que des mises à jour régulières pour
vous offrir toujours plus de choix. Rejoignez notre communauté et
profitez de la meilleure expérience de streaming VF et VOSTFR, sans aucune
limite
Amazing a lot of fantastic knowledge!
Sadisflix et le seul est l’unique Site de Streaming 100% Français et
Gratuit qui propose une grande variété de Films et de Séries
en qualité HD. Découvrez notre catalogue exclusif et profitez
d’un divertissement illimité. Nous vous offrons une sélection de contenus adaptée à vos préférences et à
vos envies, ainsi que des mises à jour régulières pour vous offrir
toujours plus de choix. Rejoignez notre communauté et
profitez de la meilleure expérience de streaming VF et VOSTFR, sans
aucune limite
Sadisflix et le seul est l’unique Site de Streaming 100% Français
et Gratuit qui propose une grande variété de Films
et de Séries en qualité HD. Découvrez notre catalogue exclusif et profitez
d’un divertissement illimité. Nous vous offrons une sélection de
contenus adaptée à vos préférences et à vos
envies, ainsi que des mises à jour régulières pour vous offrir toujours plus
de choix. Rejoignez notre communauté et profitez de la meilleure expérience de streaming
VF et VOSTFR, sans aucune limite
Thanks a lot, Good information!
You suggested it exceptionally well.
Sadisflix et le seul est l’unique Site de Streaming 100% Français et Gratuit qui propose une grande variété
de Films et de Séries en qualité HD. Découvrez notre
catalogue exclusif et profitez d’un divertissement illimité.
Nous vous offrons une sélection de contenus adaptée à
vos préférences et à vos envies, ainsi que des mises à jour régulières pour vous
offrir toujours plus de choix. Rejoignez notre communauté et profitez de
la meilleure expérience de streaming VF et VOSTFR, sans aucune limite
You said it very well..
Вы истинно отлично полагаете. Прекрасно, что я сюда пришел. Благодарность!
Many thanks! I value it.
You actually stated this wonderfully!
Just wish to say your article is as astonishing.
The clarity to your post is just great and i can think you’re an expert in this
subject. Fine together with your permission let me to grab your
feed to stay up to date with approaching post.
Thanks a million and please continue the rewarding
work.
Bonus buy slots are online slots where you can pay a premium in exchange for instant access
to their main bonus feature; usually the free spins round.
Also known as “feature buy”
You definitely made your point!
Thanks in support of sharing such a good idea, post is
nice, thats why i have read it completely
Whoa many of superb advice!
I believe people who wrote this needs true loving because it’s a blessing.
So let me give back and give true love to change your life and if you want to
with no joke truthfully see I will share info about how to change your life Don’t forget..
I am always here for yall. Bless yall!
%%
اخبار جدید خانه وحشت تهران و اخبار هنرهای تجسمی ایران و اخبار آزمون علوم پایه پزشکی و اخبار ورزشی نود
و اخبار علوم پزشکی همدان و اخبار علمی سرپوش
https://yoursocialpeople.com/story13693492/اخبار-جدید-resident-evil-4-remake
Howdy! Do you use Twitter? I’d like to follow you
if that would be ok. I’m undoubtedly enjoying
your blog and look forward to new updates.
Thank you. I enjoy this!
Whoa tons of beneficial tips!
Salut Eric. Tres sympa tes tutos sur DazStudio.
Mais faudrait vraiment que tu règle ton problème de son, c’est
très faible. Sinon continue, tu as gagné un abo
🙂
All sporsbooks are distiinctive when it comes to deposit bonusees and loyalty
applications.
Fantastic goods from you, man. I’ve understand your stuff previous to and you’re just
extremely fantastic. I really like what you have acquired here,
certainly like what you’re saying and the way in which you say it.
You make it entertaining and you still care for to keep it sensible.
I cant wait to read far more from you. This is actually a terrific website.
lordfilm сериалы
I have learn a few just right stuff here. Certainly value
bookmarking for revisiting. I surprise how so much attempt
you put to make this kind of fantastic informative
website.
Good day! Do you use Twitter? I’d like to follow you if that would be okay.
I’m absolutely enjoying your blog and look forward to new updates.
Do you mind if I quote a couple of your articles
as long as I provide credit and sources back to your site?
My website is in the exact same area of interest as yours and my visitors
would certainly benefit from some of the information you provide here.
Please let me know if this ok with you. Cheers!
Good day! Do you use Twitter? I’d like to follow you if that would be okay.
I’m definitely enjoying your blog and look forward to new posts.
Great delivery. Sound arguments. Keep up the great effort.
Hi there, I enjoy reading through your article. I like to write a little comment to support you.
Regards, Wonderful stuff!
abc slot gacor hari ini buktikan sekarang
https://dpm-ptsp.gorontalokab.go.id/tintuc/ezyzpv
Fine posts Thank you!
Discover the iconic San Juan Fort or wander by way of
the street for unexpectted spots.
Appreciate it! A good amount of write ups!
With thanks. I like it.
Thanks. Fantastic stuff!
Nicely put, Thank you!
Amazing tons of awesome advice!
With thanks. Numerous facts.
muha meds cart price
Good content Thanks a lot.
Very descriptive article, I loved that a lot. Will there
be a part 2?
To do this, it’s important to take the drug as your doctor directs, and to not skip doses.
menara368
menara368
Ich wollte einfach einen netten Gruss da lassen. Bin eben auf die Homepage gestossen.
https://rowanhphl07407.ampblogs.com/planet88-login-planet88-mudah-menang-terjamin-gacor-new-member-55093780
I’d like to find out more? I’d love to find out more details.
Supreme Court ruling overturning a 1992 law that barred it in most areas.
You said it very well..
Watching this, thought of another way. Create new surface from the region you want to hide and set opacity of it to 0.
Will it work? In batch render?
Fine knowledge Many thanks!
There’s certainly a great deal to know about this issue.
I like all of the points you have made.
If you are going for best contents like I do, only pay a quick visit this
web site every day because it offers feature contents, thanks
Information nicely used!.
یک خرید
You stated it wonderfully.
Fine tips, Thanks.
Good data, With thanks!
Genuinely when someone doesn’t understand afterward its up
to other visitors that they will assist, so
here it takes place.
I’ve been browsing online greater than 3 hours these days,
yet I by no means discovered any attention-grabbing
article like yours. It is lovely worth enough for me.
In my view, if all webmasters and bloggers made just right content as
you probably did, the internet might be a lot more helpful than ever
before.
Cheers. Ample stuff!
You said this very well.
Terrific forum posts, Cheers.
Great article. I am going through some of these issues
as well..
This post is a great resource for anyone looking to improve their knowledge on the topic.
Helpful advice, Thanks!
Lovely material, Thanks a lot.
фильмы LordFillm (unleashwithin.com)
Alo Print Đội ngủ mẫu mã và in ấn túi giấy trên 10 năm kinh
nghiệm . địa chỉ ngay để được trả lời.
In túi giấy cho những cửa hàng quần áo thời
trang , mỹ phẩm, quà tặng nhẹ đẳng cấp,
lịch sự. tư vấn hết dạ. ngoài mặt Theo đề xuất.
Free ship nội thành.
Thanks a lot. Loads of data.
Many thanks! I like it.
Regards, Great information.
I’m truly enjoying the design and layout of your website.
It’s a very easy on the eyes which makes it much more enjoyable for me to come here and visit more
often. Did you hire out a developer to create your theme?
Superb work!
The mobile app makes it possible for players to access their account from anywhere in the planet.
сасалкино
Thanks a lot, Wonderful stuff.
Amazing information Thanks.
This is nicely said. !
You’re so awesome! I don’t think I have read a single thing
like that before. So great to find someone with some genuine thoughts on this subject.
Seriously.. many thanks for starting this up. This site is
something that is needed on the internet, someone with a
little originality!
Cheers. Lots of material.
Thanks. Awesome stuff.
Alo Print Đội ngủ thiết kế và in ấn túi giấy trên 10 năm kinh
nghiệm . liên hệ ngay để được tư vấn. In túi giấy cho các shop xống áo phong cách , mỹ phẩm, quà tặng nhẹ
đẳng cấp, lịch sự. trả lời nồng nhiệt.
bề ngoài Theo bắt buộc. Free ship nội thành.
Amazing info, Thanks.
Despite the fact that tooth staining is one of chlorhexidine’s unplanned side effects, it is still reliable in dealing with gingivitis. If you are certain that the staining is triggered by the foods or beverages you eat, there is an easy natural remedy that can frequently reverse it. Brush your teeth with cooking soda and also water every few days to supplement your usual oral care regimen. LISTERINE ULTRACLEAN ® DISCOLORATION PROTECTION Antiseptic Mouthwash can aid maintain these discolorations away from your mouth.
Tooth sensitivity takes place when the internal layer of your tooth, known as dentin, comes to be exposed. This kind of toothache happens even when there”s no tooth cavity to find. Dentin normally ends up being exposed when there”s a wearing off of enamel or periodontal recession.
A cracked tooth, if not treated quickly, can cause better fractures, as well as the soreness will gradually boost. The best treatment is for a skilled dental professional to do an origin canal, as the signs can worsen in time, as well as the discomfort will certainly boost. When your youngster loses their initial collection of teeth, you’ll observe severe level of sensitivity as the tooth becomes looser. This is particularly pronounced when you attack down as the nerve ending underneath the tooth is stimulated.
Blockage Or Sinus Stress
If the tooth harms when you bite down, after that it could be a sign for a hidden condition. Allow’s look at some reasons for why you encounter tooth pain while biting. In an oral context, occlusion means the call in between your teeth. Simply put, occlusion can describe the way your teeth bite with each other. Ideally, the stress your mouth puts in while attacking down must be distributed evenly. This suggests that no specific tooth or filling is bearing too much force than it should
Today, dental experts are able to use their patients easy remedies that can be used in the workplace or in the house. A dental expert can eliminate plaque and tartar off the surface area of the teeth, as well as in between the teeth with a manual or ultrasonic scaler. There are additionally natural home remedy like cleaning teeth with soft drink and also water. Surface area spots might be feasible to remove with at-home whitening procedures. Nonetheless, whitening procedures might not help all tooth stainings. Before teeth whitening, an individual’s teeth as well as gum tissues are meticulously cleaned, adhered to by the application of a bleaching representative.
If staining is brought on by tartar or clinical problems, an individual might require to go to a dental professional. While signs and symptoms might resemble those of tooth decay, fluorosis is generally harmless. This problem is brought on by a disruption in development which causes enamel to be difficult however slim.
This stick indicates tooth pain at certain points when you apply stress. If your tooth harms with pressure put on it, you intend to temporarily prevent chewing on that particular side of your mouth. Tidy around your gums well to find out if something is wedged in between your teeth. Nonetheless, if your signs and symptoms linger for greater than a couple of days, you’re most likely past the factor of attempting to relieve the tooth pain by yourself. Are you struggling with tooth pain or teeth sensitivity after eating? You might really feel tooth discomfort after eating certain sorts of food.
The rinsing will lift away some of the stain fragments before they “settle” right into your teeth. If you ingest excessive or insufficient fluoride throughout tooth advancement, it can trigger hypermineralization or hypomineralization in your enamel. Consequently, it leaves white and also brownish spots throughout the various tooth surfaces. It usually results from an effect on the tooth and worries the incisors or dogs.
Just How To Tidy Brown Spots In Between Teeth
Any time you put these dark products in your mouth or inhale them over and also over, it triggers the discolor fragments to permeate deep into the small pores/tubules of your teeth. As well as your garments and also the paint and also furnishings of your residence. Inherent tarnish is tooth staining that is within your tooth makeup. It’s not like build-up outside of your tooth that your hygienist or dental professional would certainly wipe or gloss. Yet normally, you’re not visiting a brownish or black place from a tooth cavity unless the decay is currently truly huge as well as intrusive.
The only feasible treatment to maintain a sore tooth is origin canal therapy. Serious dental caries can aggravate the nerves inside the tooth, causing pain whenever you chew, attack, or consume food. The most effective therapy for a sore tooth is to clean the contaminated nerve, after that seal the canal off with a root canal.
Need A Pediatric Dental Practitioner In Falls Church? Tips To Locate The Ideal Pediatric Dental Professional In Drops Church, Va
A not successful root canal treatment might likewise become a dental abscess. Fractured toothcan be challenging to diagnose since there may not be any signs besides discomfort when chewing. It helps to focus as to whether the discomfort is coming from a particular component of your mouth or a certain tooth.
Amazing knowledge, With thanks.
Nicely put, Thanks a lot.
Nicely voiced certainly. !
Having read this I believed it was really enlightening.
I appreciate you spending some time and energy to put this information together.
I once again find myself spending a significant amount of time both reading and commenting.
But so what, it was still worth it!
We identified nicely-becoming scores are
higher for those who are employed compared to these
who are unemployed.
Nicely put. Kudos!
Great information, With thanks!
Superb material, Thanks!
Useful write ups, Thank you!
Howdy! I’m at work surfing around your blog from my new apple iphone!
Just wanted to say I love reading through your
blog and look forward to all your posts! Carry on the superb
work!
If you are going for best contents like me, only visit this
website all the time because it presents quality
contents, thanks
Anubhav Oberoy The most beneficial Company enterprise
Engineering Cloud Technique Instructing is furnished by only one
and only mentor Anubhav Oberoy Apparent and new new inspect substance and modern-day information are created accessible for all learners.
sap ui5 education
If you’d like to sign up for Reside batch of Anubhav of SAP Cloud Platform Schooling , Cloud Application Programming Product or
service – CAPM Then you can certainly consider http://www.anubhavtrainings.com and register all all on your own for This
method.
You actually suggested that very well!
You suggested this perfectly.
I’m not sure why but this site is loading very
slow for me. Is anyone else having this issue or is it a problem on my
end? I’ll check back later and see if the problem still exists.
Local business have the advantage of offering tailored solutions to their customers. Bigger providers accommodate a broader audience as well as have to stay on top of the needs of the masses. In contrast, small businesses can easily pay attention to the individual demands of their consumers. A little grocery store may create sure they keep their client’s preferred label of grain in sell, or even a regional suit maker may create changes to the specific spec of their consumers. Through offering tailored services, small companies can easily build outstanding customer connections and also use a distinct consumer expertise, https://www.tumblr.com/emma-thyst/718448208427237376/the-six-things-you-need-to-consider-before?source=share.
Nice answers in return of this query with solid arguments and explaining everything regarding that.
Regards! Useful stuff!
Wonderful facts, Thanks!
Helpful tips, Regards!
Thanks, Plenty of tips!
Thanks! I like it!
You said it terrifically!
You actually said this terrifically!
Point effectively applied..
To promote to tthe final level of this part-time job, players will have to have their Sim
reach level 2 of the Gardening Talent.
You’ve made your point extremely well!!
Awesome data, Thanks!
Hi there! I’m at work browsing your blog from my new iphone
4! Just wanted to say I love reading through your
blog and look forward to all your posts! Carry on the
excellent work!
Fine tips, Thanks!
Great article, just what I needed.
Hi! Do you use Twitter? I’d like to follow you if
that would be ok. I’m absolutely enjoying your blog
and look forward to new updates.
Ce contenu intéressant m’a vraiment interpellé.
En tant que négociateur chevronné, j’accorde
une grande importance à la performance commerciale. J’ai trouvé beaucoup de contenu sur les logiciels
de vente IA qui semblent vraiment optimiser les processus de vente.
해킹 피해 발생시 해킹 피해 해결을 위한 전문가가 상담을 해드립니다.
중요한 디지털 자산을 지켜드리고 나아가 미래에 발생할
수 있는 위험 리스크가 생기지 않게 보안의식을 가질수 있게 안내해드립니다.
또한 포렌식 디지털 증거 수집 및
복구 등, 바이러스 검수 등 업무도 하고 있습니다.
참새왕 해킹 의뢰 서비스
I believe people who wrote this needs true loving because it’s a blessing.
So let me give back and speak out on change your life and if you want to with no joke truthfully see I will share info about how to make passive
income Don’t forget.. I am always here for yall.
Bless yall!
Kudos! Terrific stuff!
Nice response in return of this issue with genuine arguments and
explaining the whole thing regarding that.
You actually revealed that superbly!
This is nicely expressed. .
Félicitations pour cet article captivant !
L’IA dans les logiciels CRM représente une avancée majeure pour les professionnels.
Good day! This is my first comment here so I just wanted to give a quick shout out
and say I truly enjoy reading your posts. Can you suggest any other blogs/websites/forums
that go over the same subjects? Thanks for your time!
I’d like to find out more? I’d like to find out some additional information.
Hi there! I’m at work surfing around your blog from my
new iphone 3gs! Just wanted to say I love reading through
your blog and look forward to all your posts! Keep up the excellent work!
Wonderful information. Thanks.
What is an example of quality assurance fildena 120 mg
Beyond the handful oof actors that headline any dramaa or film,
there are hundreds additional that deal with scripts, makeup, lighting,
and more.
Terrific stuff, With thanks!
Увеличение связи между щенком и владельцем: дрессировка помогает укрепить отношения между владельцем и питомцем. Такая дрессировка не помогает щенку развиваться, а, наоборот, портит его. Это называется “дрессировка на обратный результат”. Это развлечет вашего бигля, а также облегчит его боль. Когда малыш находится в очередном процессе поедания вашего носка, командным тоном произнесите «Фу». Когда щенок начинает реагировать на своё имя, значит, он уже понимает, что хозяин обращается к нему и что-то от него хочет. По следам на железной ловушке, в которой погиб щенок его и Лизы – служебной собаки пограничников, Чёрный волк понимает, что Франек убил щенка и подставил его хозяйку при незаконном переходе границы, и нападает на лесоруба. По возвращении в Вену Бенни вдруг обнаруживает, что Кайнц, который отныне скрывается от полиции, посещает один из венских тиров. Итальянский комиссар Лоренцо Фаббри, приехавший в Вену по делу, привязывается к Рексу и забирает его с собой в Рим. Однако во время очередного дела он был ранен, и от смерти его спас комиссар Марко Терцани, назначенный на место Риверы.
Appreciate the recommendation. Let me try it out.
I am genuinely thankful to the holder of this site who has shared this impressive
paragraph at here.
лордфильм
I believe people who wrote this needs true loving because it’s a blessing.
So let me give back and show back my secrets on change
your life and if you want to really findout?
I will share info about how to make a fortune Don’t forget..
I am always here for yall. Bless yall!
You reported that well!
Regards, I like it.
What’s Going down i’m new to this, I stumbled upon this
I’ve discovered It absolutely helpful and it has aided me
out loads. I am hoping to contribute & aid different customers like its aided me.
Good job.
Hello there! This is my first comment here so I just
wanted to give a quick shout out and tell you I really enjoy reading through your posts.
Can you suggest any other blogs/websites/forums that deal with the same
topics? Thank you!
I believe people who wrote this needs true loving because it’s a blessing.
So let me give back and give out my secrets to change your life and if you want to seriously get to hear I will share info
about how to learn SNS marketing Don’t forget..
I am always here for yall. Bless yall!
Useful data Thanks a lot.
Cheers, Plenty of facts.
You have made your point!
With thanks. Excellent stuff!
Hey there! I’m at work browsing your blog from my new apple iphone!
Just wanted to say I love reading through your
blog and look forward to all your posts! Keep up the
fantastic work!
I do not even know how I ended up here, but I thought
this post was good. I do not know who you are but definitely you are going to a famous
blogger if you are not already 😉 Cheers!
Fine posts, Regards.
SLOT GACOR
Slot Gacor Gampang Menang : Gate Of Olympus , Starlight Princess, Koi Gate , Bonanza
Provider Slot Online Terbaik : Pragmatic Play , PG Soft
Promo Unggulan : Event Scatter , Petir Merah , Bonus Begadang 78%
Pembayaran : BCA , BRI , BNI , Mandiri
E-Wallet : Dana , Ovo , Gopay , Pulsa
Minimal Deposit : Rp 25.000,-
Excellent info, Thanks!
Rhinocort nasal spray is unsuitable for some patients.
Two of the studies evaluated patients at remedy centers within the United States, one came about in Turkey and the
opposite included 71 medical centers in 14 countries.
Research members, who underwent X-rays or nasal endoscopy to verify prognosis, received either a placebo or
intranasal corticosteroids for 2 or three weeks, alone or together with
antibiotics. Budesonide is an anti-inflammatory drug
that belongs to a bunch of medications called corticosteroids.
5. To keep away from complications in a symptoms’ resumption after steady drug use duration should not exceed
7 days. Typically, it’s endorsed to make use of Rhinocort one or two occasions per day –
it ought to be four sprays into each nostril day by day,
so you may take this dose all within the morning,
or you can separate it into 2 sprays in each nostril
in the morning and night. Tilt your head to the facet and pour the solution inside one nostril.
Negative effects are generally mild and will embody nosebleed, sore throat, nasal irritation, and cough.
Itulah seputar panduan disaat akan bergabung menjadi member
di situs slot online kami ini yang pastinya sangat membantu para pemain baru maupun pemain lama yang akan bermain lagi dan juga Anda tidak akan merasakan rugi disaat bergabung bersama kami dikarenakan situs kami juga memiliki update update terkait bonus bonus dan juga permainan permainan yang baru pastinya agar Anda merasa nyaman dan puas terhadapa situs kami ini.
Disaat Anda akan bermain slot online pastinya pendaftaraan sangatlah penting dikarenakan agar Kalian tercatat sebagai member situs slot online tentunya.
SLOT GACOR
Slot Gacor Gampang Menang : Gate Of Olympus ,
Starlight Princess, Koi Gate , Bonanza
Provider Slot Online Terbaik : Pragmatic Play ,
PG Soft
Promo Unggulan : Event Scatter , Petir Merah , Bonus Begadang 78%
Pembayaran : BCA , BRI , BNI , Mandiri
E-Wallet : Dana , Ovo , Gopay , Pulsa
Minimal Deposit : Rp 25.000,-
Ao conhecer o jogo, e que o sucesso depende da habilidade do jogador em jogar o jogo. Qual é o limite máximo de retirada no Fish Eye? Jogar caça-níqueis pode ser emocionante e divertido, você pode até mesmo sair com mais dinheiro do que entrou.
Thank you! Quite a lot of facts!
You have made the point.
I’m truly enjoying the design and layout of your site. It’s a very easy on the eyes
which makes it much more pleasant for me to come here and visit more often. Did
you hire out a designer to create your theme? Superb work!
Cheers! I enjoy it.
Wow tons of amazing info.
slot gacor, situs slot gacor, slot gacor hari ini, slot gacor gampang menang, link slot gacor, rtp slot gacor, rtp slot gacor hari
ini, situs slot gacor 2023, https://sipermen.jatimprov.go.id/-/
You actually said it fantastically!
фильмы lordfilm
Nicely put, Cheers!
Кто либо знает, каким способом контактировать с админом? Катаю в обратную связь, он не дает ответ!
Excellent knowledge, Thank you.
Game slot terbaik dengan rtp di atas 90 persen seperti
koi gate, fa cai shen dan hot hot fruit siap berikan keuntungan besar bagi para pemainnya.
Namun sekarang saat nya kita mengetahui 6 provider slot gacor Slot Online yang paling populer saat ini.
Sudah populer di Asia adalah title untuk
jenis game situs satu ini yang bernama PgSoft. Terdapat ratusan jenis slot online resmi didalam PgSoft
yang sudah pasti menguntungan sekali untuk Anda para pecinta judi slot online.
Sebelum berkembangnya situs slot online, J88SLOT sudah ada dimesin slot
online yang kita kenal dulu seperti permainan dingdong pakai
koin dengan tombol spin.
But the 22 civil lawsuits are proceeding and may well not
go to trial until next year.
You mentioned it wonderfully!
Fine info, Kudos!
Kudos! Lots of info.
фильмы лордфильм
Regards. Great information.
When thyroid gland doesn’t function correctly, it will probably induce adjustments in total energy expenditure, physique weight, and composition, and the body’s temperature, the proof says.
#VernonWells
As Ukraine presses south after liberating the Kharkiv region in the north last month, authorities in the western Russian provinces bordering northeastern Ukraine appeared jittery.
I enjoyed your article KSNl
You made your position extremely well.!
You made your stand extremely nicely!.
Negara emg tidak punya uang corona aja dari hutang, negara tetangga keluar 1000t cuma pakai dana cadangan.
ARL Commission chairman Peter V’landys stated the league may ‘not guarantee the security of players’.
The Clip-Tone gadget might be a easy resolution.
An intriguing discussion is definitely worth
comment. I do think that you ought to publish more about
this topic, it may not be a taboo subject but generally people do not talk about these subjects.
To the next! All the best!!
INCS stands for intranasal corticosteroid nasal spray.
A verify to see if the drainage contains beta-2 transferrin proves or
disproves the cerebrospinal-fluid trigger.
You definitely made the point.
Even if the node operators understand what they are doing, their users and
their users’ friends who are invited to join The New Secure Facebook are not capable of evaluating their
security on Diaspora Anonymous’s most high-profile hack
in the.
Новости в telegram
love the ass
Hello there! This is my first comment here so I just wanted to give a quick shout out and say I genuinely enjoy reading your
posts. Can you recommend any other blogs/websites/forums
that deal with the same topics? Thanks a ton!
I’m not sure why but this site is loading very slow for
me. Is anyone else having this problem or is it
a issue on my end? I’ll check back later on and see if
the problem still exists.
Akibatnya, Anda mengantongi kemampuan menjumpai memenangkan jackpot besar, kecil, pula besar melalui budi ini. Hanya serta persediaan sedimen minimum sebesar Rp20.000 Anda dapat bermain dan mengibarkan taruhan di alat slot idola Anda. Bahkan selain itu pun vital kepada Kamu bisa berburu aktiva arta asli Rp10.000 mulai bertindak permainan tersebut. Anda mengenai menerima proklamasi transmisi arta saldo bermain telah sukses. Roulette sendiri dimainkan melalui sebuah Wheel berisi digit-skor yang berkenaan diputar berbarengan menggunakan bal kecil nan dilemparkan berlawanan mata angin adegan Wheel. Dalam kira-kira pertunjukan slot, ada saja karakter premi yang memicu fitur khusus sepantun mini-games ataupun putaran tambahan kepada membantu Anda meningkatkan kejayaan Anda. Dengan adanya kondisi ini, tentu terhadap mengeluarkan Anda semakin kerasan lagi makmur selama terus main-main dan membaca keuntungan. Proses endapan mau dibantu sama layanan customer support sepanjang 24 weker. Bocoran format slot gacor menjadi lengah esa punat kabar terutama nan tentu diketahui akibat panggar player posisi gambling slot online Hoki178. Tidak kalah berguna rekomendasi terbaik yang saat ini sedang terkenal dimainkan merupakan pragmatic play karena hadirin maka pelaku terbanyak saat ini menjadi rekomendasi terbaik awak dapat bermain slot online mulus menang oleh menggunakan lokasi slot gacor onlien dana asli.
Tangkap para bandit itu dan dapatkan hadiah terbesar di TIKTOKSLOT88.
Jika Anda berhasil menangkap mereka, hadiah uang 30 juta
akan menjadi milik Anda. Menangkan lagi pengganda
dan simbol bingkai emas yang berubah menjadi simbol liar.
Wild Bandito adalah game slot 5 gulungan dan 4 baris yang bertema simbol bingkai emas dan spin gratis dengan pengganda yang terus meningkat.
Selama mode spin gratis, semua simbol kecuali simbol liar dan simbol scatter di gulungan 3 akan muncul sebagai
simbol bingkai emas, meningkatkan kesempatan anda untuk menang jackpot!
Dapatkan fitur spin gratis dan menangkan pengganda dengan 4 simbol scatter.
You actually said that effectively!
Thanks! Great information!
Can you tell us more about this? I’d love to find out more
details.
Great knowledge, Thank you!
Tips well utilized.!
Information well used.!
http://cctvsmart.ir
We are glad you love GraphicMama’s Character Animator puppets and
really appreciate your positive feedback. Cheers!
Excellent write-up. I absolutely love this site.
Stick with it!
Investors should make an independent judgement as to whether trading is appropriate for them in light of their financial condition, investment experience, risk tolerance and
other factors.
Thanks! Plenty of knowledge!
Either way, most will be compatible with IOS and Android devices – hence allowing you to play froim tablets and
mobile phones.
Let’s Optimize your Marketing. There is NO link to ANY landing page in your description.There is
no way to CONTACT you here???
Amazing tons of wonderful info.
Asking questions are actually fastidious thing if you
are not understanding anything entirely, except this article provides good understanding even.
Useful material, With thanks.
Your means of explaining the whole thing in this article is
genuinely nice, every one can simply know
it, Thanks a lot.
I’m gone to say to my little brother, that he should also visit this
web site on regular basis to get updated from most recent gossip.
Kudos, Loads of info!
Seriously all kinds of wonderful data!
Appreciate it. Numerous forum posts!
русская порнуха
Happy Travels
Cheers, Plenty of content.
You’re so interesting! I don’t believe I have read a single thing like this before.
So nice to find another person with a few unique thoughts on this topic.
Seriously.. thank you for starting this up. This
web site is one thing that’s needed on the web, someone with some originality!
Hello, i believe that i noticed you visited
my website so i got here to go back the favor?.I
am trying to to find things to improve my website!I assume its good enough to use some of your concepts!!
Very inspiring, Renata! Great job.
Really plenty of very good knowledge.
You could definitely see your skills within the article you
write. The world hopes for more passionate writers such
as you who aren’t afraid to say how they believe. At all times follow your heart.
Nicely put. Thank you.
Wow tons of useful tips!
Can I show my graceful appreciation and shout out on really good stuff and if you want to have a checkout Let me tell you a brief about howto make
money I am always here for yall you know that right?
Some genuinely interesting information, well written and broadly user friendly.
Amazing content Appreciate it!
I was able to find good information from
your articles.
Superb content, Many thanks!
Nicely put. Cheers.
Hi there! Do you use Twitter? I’d like to follow you if that
would be okay. I’m undoubtedly enjoying your blog and
look forward to new posts.
i jus tlove this moivei iawsy wanrted to stay at the royal suit in the
plaza hoitel it is defently on my bucket list ALONG WITH THE PRESDENCHEL ST REGES SUIT IN NEWYOIRK CIUTY
Thank-Like ปั้มไลค์ฟรี อันดับ 1
Have you ever thought about including a little bit more than just your articles?
I mean, what you say is fundamental and all. Nevertheless think of if you added some great visuals or
videos to give your posts more, “pop”! Your content is excellent but with images and clips, this blog could definitely be one of the
greatest in its field. Great blog!
This is nicely said. .
Thank you, Plenty of tips.
slot gacor, situs slot gacor, slot gacor hari ini, slot gacor gampang menang, link slot
gacor, rtp slot gacor, rtp slot gacor hari ini, situs slot gacor 2023, https://sipermen.jatimprov.go.id/-/, Daftar Situs Slot Gacor Hari Ini
Dan Terbaik Terpercaya Gampang Menang Maxwin 2023
Appreciate the recommendation. Let me try it out.
Great write ups, Kudos.
The author is known by the name of Octavio Winchester.
Doing aerobics is the hobby he will probably never stop doing.
Invoicing is where his primary income was produced from but he’s always
wanted his own small business. New York is where our home is.
What’s up to every one, since I am truly eager of reading this
webpage’s post to be updated regularly. It contains pleasant stuff.
5 days.
Great article, totally what I needed.
Thanks! A lot of tips.
She had nno sick leave and had to quit her job as a waitress and bartender.
Thank you, I like it!
壹葡娛樂/壹博娛樂
519 Lockhart Rd, Causeway Bay
+85292121530
https://onebets.org/
You said it very well..
Now Tv viewers are watching their favourite applications on their laptop screens. I have a whole lot of Indian viewers on YouTube and once more, as with AdThrive, I’m certain sure international locations are extra profitable than others. I have not got any concrete plans but you are looking at what merchandise you could possibly have, maybe bodily ones, apps or there’s a variety of ebooks around. I feel I was actually having fun with doing so I had like a Kids Get Arty series, taking a look at Goldsworthy or Andy Warhol or whatever. I don’t wish to be advertisements to be intrusive obviously however equally, I believe if they’re high-quality. What would you say to somebody who involves you and says “I wish to do what you do. I would like to start out something by myself”? By November 2001, the TSA was rolling out a number of latest safety measures: armed air marshals, bolstered cockpit doorways and no-fly lists identifying individuals who might pose a threat and designating them for enhanced screening or, as applicable, prohibiting them from boarding an aircraft.
How do online slots work – https://forbrugermania.com
https://forbrugermania.com
%%
Wow plenty of useful tips!
Amazing many of useful material.
Nicely put. With thanks!
You have made your point extremely well.!
Reliable tips Many thanks.
Regards. An abundance of data.
Cheers, Lots of information!
Thanks a lot. Awesome information.
Reliable data Thank you.
This paragraph gives clear idea in support of the
new people of blogging, that genuinely how to do
blogging.
We all understand the value of a good night’s sleeping for our bodily wellness, yet did you know that it additionally participates in an essential job in boosting your cognitive feature? Your brain works relentlessly throughout the day, refining relevant information, choosing, as well as holding memories. Just like your physical body, it needs to have time to remainder and recharge, http://sqworl.com/mgilm2.
Thanks a lot. Quite a lot of knowledge!
Regards, A lot of postings!
Wow lots of excellent facts!
Whoa a good deal of excellent material.
Wow a good deal of very good tips.
I visited several sites but the audio feature for audio
songs existing at this site is actually fabulous.
A lot may probably take place when you’re on the road. You might face an unforeseen accident, lose energy, obtain a puncture, or your vehicle’s battery may give up. You can not merely stay where you are actually, waiting for a miracle to happen, https://anotepad.com/notes/8yixp97n.
You definitely made your point!
You stated this effectively.
Really many of very good data.
Regards. Plenty of stuff!
Terrific data. Thank you.
None of them require you to be registered to start a
random chat.
It’s a pity you don’t have a donate button! I’d most certainly donate to this superb
blog! I suppose for now i’ll settle for book-marking and
adding your RSS feed to my Google account. I look forward to fresh updates
and will talk about this blog with my Facebook group.
Chat soon!
Superb content. Regards.
Thanks a lot, An abundance of write ups.
Tips very well taken!.
With thanks! Plenty of posts.
Nicely put. Appreciate it.
Thank you, I appreciate it!
Awesome content, Thanks.
Examples of small-scale wind power projects in an city setting will be present
in New York City, where, since 2009, a number of
building initiatives have capped their roofs with Gorlov-sort helical wind turbines.
They will cater the load demand perfectly and work as base load power plants when the extracted photo voltaic power
is found excess in a day. In mild load conditions, where the loading on transmission lines is nicely
beneath the surge impedance loading, the effectivity of the power system may actually be improved by switching in reactors.
Photovoltaic cells convert mild into an electric present using the photovoltaic impact.
Electric power will be delivered over lengthy distances by transmission traces and used for functions reminiscent of
movement, light or heat with excessive effectivity.
2019) found that Chinese language city-dwellers could also be resistant to constructing wind turbines in city areas,
with a surprisingly high proportion of individuals citing an unfounded worry of
radiation as driving their issues. Although wind power is a well-liked form of energy era, onshore or close
to offshore wind farms are generally opposed for his
or her influence on the landscape (particularly scenic areas, heritage areas
and archaeological landscapes), in addition to noise, and impression on tourism.
Good day! Do you use Twitter? I’d like to follow you if that would be okay.
I’m undoubtedly enjoying your blog and look forward to new updates.
Dengan mengikuti tips-tips di atas, Anda dapat meningkatkan peluang menang dan memaksimalkan pengalaman bermain Anda di Yoi4D.
Semoga sedikit banyak tips dari kami bisa menjadi berguna untuk kalian para pemain slot deposit
pulsa terpercaya.
3. Tie a knot at one end of a long piece of yarn (sorry, I don’t have an exact length – it depends on the size of your box) and run the yarn through the holes, from one side of the box to the other. 2. Run the yarn from one side of the warp to the other, going under and over the warp yarn. If you have five dangling warp yarns at each end, you should tie a total of five knots. 1. When the weaving is done, make a know of the weft to the warp. 1. Place another popsicle stick at the other end of the weaving to straighten it out. 4. Your weaving is done! I’ll go out on a limb and assume that we all know that corrugated boxes start their life as a tree. I’ll share all the steps here. This giant castle is perfect for a lot of activities. If you are transporting your castle later, hold the pieces together using binder clips and or clothespins. Cut several pieces in different shapes, color them, poke holes, and use fluted dowel pins for guiding the different pieces. 6. With a sharp box cutter, cut out a door, windows, and whatever else you want to make your cardboard castle.
Corrugated box – https://kardus.co.id
https://kardus.co.id
You reported this really well.
Je tiens à exprimer toute ma gratitude au webmaster de
ce site pour le partage de ce contenu captivant. Votre générosité
à partager vos connaissances sur la relation client et
les logiciels CRM est très appréciée. Vos articles ont été
une source d’inspiration pour moi et m’ont permis d’approfondir mes
connaissances dans ce domaine passionnant.
Helpful data Cheers.
Many thanks! A lot of content!
Good day! Do you use Twitter? I’d like to follow you if that would be ok.
I’m absolutely enjoying your blog and look forward to new updates.
Many thanks. I appreciate it.
walmart pharmacy prices http://www.pharmacymaxcare.com
Good day! Do you use Twitter? I’d like to follow you if that would be okay.
I’m absolutely enjoying your blog and look forward to new updates.
Hamid Μіr M.D. Spine Surgeon Newpirt Beach
3501 Jamboree ᎡԀ #1250,
Newport Beach, CA 92660, United States
+19499916451
qualified spine surgeon cost
I’d like to find out more? I’d love to find out more details.
Appreciate it. Loads of data.
Excellent write-up. I definitely appreciate this site. Continue the good work!
Hi there! Do you use Twitter? I’d like to follow you if that would
be okay. I’m undoubtedly enjoying your blog and look forward to new posts.
I’m really enjoying the design and layout of your website.
It’s a very easy on the eyes which makes it much more enjoyable for me to come here
and visit more often. Did you hire out a developer to create your theme?
Superb work!
Be sure to see your dental expert frequently for cleansings as well as evaluations. Along with nipping any kind of dental issues in the bud, it is very important to develop a partnership with a normal oral practice. By doing this, you are in the system, so in case of a dental situation, you can take advantage of emergency situation advantages with your dental professional.
can be turned around by taking actions toward great oral health. Throughout early demineralization, direct exposure to fluoride, daily brushing as well as flossing, and regular cleanings can all aid stop & #x 2014; or perhaps reverse & #x 2014; dental cavity.
The acids in plaque liquify the enamel surface of the teeth, creating holes in the teeth called dental caries. Dental sealants are an excellent option for children whose teeth are at risk to cavities and also decay. When a fluoride varnish is placed on the teeth, it will certainly stop the microorganisms from multiplying as well as developing the enamel on the teeth. This assists to make the teeth a lot stronger, which stops future cavities.
Brown places on the teeth are common among individuals with gastric condition, especially children. Old dental repairs can additionally cause surface discolorations, making the teeth appear brownish. The majority of dental fillings, crowns, and also bridges will eventually lose their shade. When you do not get rid of plaque from your teeth on a regular basis, it can harden right into tartar. Tartar can differ in color from yellow to brownish and develops along the gum tissue line.
GLO, Snow, and also AuraGlow are effective as well as credible LED teeth whitening package brands. You need to use them continually to achieve the wanted results. However, make certain not to overuse them as they may influence tooth enamel as well as increase sensitivity. The enamel that covers the tooth’s external layer will also weaken as you age. As the acids damage the enamel, the tooth’s yellow layers listed below come to be much more visible.
Likewise, make certain to see a dentist every 6 months to obtain a thorough cleansing. Tartar buildup is most common in between teeth as well as along your gum line. The tartar itself is porous and also is highly at risk to discoloration as well as discoloration. Foods, drinks, as well as various other variables can turn it an obvious brownish color therefore. They might likewise suggest health and wellness concerns, like celiac illness. As we age, the enamel of the teeth begins to wear away, subjecting the underlying layers of yellow dentin as well as causing yellowish-brown areas.
Utilize a cotton bud to use a couple of drops of the oil directly on the spot of the tooth dental caries and allow it to soak in. Clove oil has actually been made use of commonly before the modern-day breakthroughs of oral methods to treat both the discomfort of a cavity and to restore enamel. The oil creates a numbing effect to deal with pain and also can aid your tooth retain minerals.
How Does Drinking Water Prevent Dental Caries?
Our site is not intended to be an alternative to expert medical advice, diagnosis, or treatment. Tidy between your teeth daily with dental floss or interdental cleansers, such as the Oral-B Interdental Brush, Reach Stim-U-Dent, or Sulcabrush. Brush your teeth at least two times a day with a fluoride-containing tooth paste. Ideally, brush after each dish as well as specifically before going to bed.
If the decay has actually produced an opening in the tooth’s enamel yet has actually not infected the tooth’s nerves, your dental practitioner will likely make a filling. After the cavity is cleaned, the dental expert positions a loading to secure up the hole in the tooth and also avoid additional degeneration. The reality is that the only method to eliminate tooth cavities is via correct therapy from a qualified dental expert. Although you can “reverse” a cavity by taking on an excellent dental health regimen, the degeneration and disintegration of the tooth are irreversible.
UI Wellness UIC’s seven health and wellness sciences colleges and health care delivery venture. It is popular that smoking and cigarette usage can create many different clinical troubles. Smoking causes condition and handicap and damages nearly every organ of the body, and also over 16 million Americans are dealing with a condition brought on by smoking cigarettes. Don’t neglect to replace your tooth brush at the very least every 3 or 4 months. Brush your tongue to remove bacteria as well as freshen your breath. Use an over-the-counter anesthetic specifically created to calm excruciating teeth.
Appropriate Brushing Techniques
Just a dental expert can deal with a tooth cavity by filling it in. Lots of oral troubles, also deep dental caries, establish without any pain or other symptoms. Routine oral checkups are the most effective method to catch a tooth cavity before it gets worse. If you’re mosting likely to eat sugar, attempt not to treat on sugary foods throughout the day. When the sugar is gone, your enamel has an opportunity to remineralize.
Howdy! This is my first comment here so I just wanted to give
a quick shout out and tell you I genuinely enjoy reading through your posts.
Can you recommend any other blogs/websites/forums that go over the same subjects?
Thanks for your time!
Nicely put, Thanks!
Seriously all kinds of awesome advice.
Fantastic material, Appreciate it.
prescription drug discounts publix pharmacy
You revealed this very well.
Best Earning Methods for Everyone
We’re extending authentication options by enabling a easy user identify / password authentication kind for many who don’t
have LDAP servers or want to have less complicated set
up. Never give out passwords or user ID info online except you recognize who you might be coping with
and why they need it. As well as to these components, some
beverages are legally required to show different information. An authorized user/utility
can request a token that encapsulates id info for his or her related entity.
We additionally added a particular widget to our dashboards to be able
to incorporate geolocations there. There’s a boy with blood group B whose
mom has blood group A. Give the possible genotypes and phenotypes of his father.
Registration kind is similar, there are only variations in the quantity and
names of input fields, so I’ll omit it right here.
The concept is to make a universally distinctive identify from
a sequence of pronounceable pseudowords – words which are
like English phrases in form however actually usually are not
real words. Just like with an Amazon bank card, rewards can only be redeemed
on Amazon. If your logs embrace IP addresses now you can visualize
them on a map.
Here are the main indications of honesty, legality and other
advantages of Ozwin Casino Sign In casino.
As an example, the Cypriot Citizenship by Investment (CBI) program calls
for a minimum investment of EUR300 ,000 in real estate or an alternative investment fund.
Likewise, the French Tech Visa offers a residence permit
to investors that straight invest or have a minimal 30 percent shareholding in a firm in France, and also work to produce or secure tasks in the
nation for 4 years. Various other remarkable programs consist of Canada’s Capitalist Visa program and Prince Edward Island’s
Business Stream.
Surplus lines insurance sold only through licensed surplus lines producers.
How much is pet insurance? Our staff is
pretty much capable of handling any lock and key problem.
Our specialized, well-trained and professional locksmith staff is near and ready to help you with your crisis, be it residential,
commercial or automotive-related. We are proud to have pet
experts in our professional customer service team and our pet blog -The Well Pet
Post, is an additional resource containing a variety of pet health and
pet behavior articles loaded with tips and information on providing pets and animals in your
care with the highest standards of health and well-being.
Our prompt response and professional locksmith services go beyond
lockout services. Call us on the phone or contact us via our website for free, we will help you
and give the best & fast towing services on time.
You can also use the form in the web site to contact us,
ask any question, and we will try to answer as soon as possible.
Hey can you make a post on wifiwazir (dot) com, becuase
it seems trending right now and i also want to
buy it.
Затем в каждую склянку пипеткой или микрошприцем добавляют термостатированные при 20 °С ДМД и н-бутанол в соответствии с табл.2.
Its such as you read my thoughts! You seem to grasp so much about
this, like you wrote the guide in it or something. I believe that you
simply could do with some percent to force the
message house a little bit, however instead of that, this is
great blog. A fantastic read. I’ll certainly be back.
Pilih Game Dengan RTP Slot Tertinggi
Yang pertama anda lakukan yaitu melacak game slot yang
memiliki RTP Slot Mulia Seperti yang telah saya tampilkan Di atas
ada banyak game slot yang memiliki RTP Live Slot dengan persentase
yang tinggi. Maka aku sarankan untuk anda menentukan salah satunya.
Lihat Bocoran Susunan Slot Gacor Pada Game Tersebut
Berikutnya jika anda sudah mengacu game slot yang
memiliki RTP Live tinggi dan anda yakin untuk memainkannya.
Maka anda dapat klik game tertulis dan akan memperlihatkan bocoran Pola Slot Gacor yang Ada Desain slot yang disediakan pastinya sudah dianalisa sebelum diberikan menjumpai anda sekalian.
Gunakan Struktur Slot Gacor Dengan Sesuai
Langkah ini ialah hal yang Terpenting karena jika anda tidak menggunakannya dengan setimpal maka bisa jadi besar struktur slot yang diperlukan akan Gagal
Maka saran kami yakni anda wajib menentukan pola slot gacor dengan sesuai untuk menjumpai
hasil yang maksimal. Banyak member yang belakangan tergelincir dan marah,
sebaliknya mereka esa yang tidak menggarap desain slot dengan sesuai.
Nice response in return of this query with genuine arguments and telling everything about that.
Hello! Do you use Twitter? I’d like to follow you
if that would be ok. I’m undoubtedly enjoying your blog
and look forward to new updates.
Fantastic write ups, Cheers.
slot Gacor Maxwin YGGdrasil
Boleh disebutkan permainan yang ada di YGGdrasil membuat pemain jadi antusiasme bermain karena mempunyai seputar games berperforma sangat baik, menyelenggarakan kreativitas kualitas terbaik di
belahan pengembang industri slot. Dengan dasar jalan yang inovatif bersama resiko
sound games yang bersih terdengan membuat Anda cicipi tiap detik permainan yang jalan.
Slot Gacor Maxwin Pragmatic Play
Kepopuleran dari games slot gacor ini tidak harus di pertanyakan kembali,
karena teregulasi dan mobile-friendly dari judi slot pragmatic
play apabila pengembang perusahaan slot teratas pada tahun 2023 ini.
slot gacor pragmatic play individual sudah mempunyai singularitas yang dikenal memberi maxwin terbaik untuk sekian banyak
pemain slot di Indonesia.
Slot Gacor Maxwin Habanero
Timbulnya games Koi Gate apabila telatah dari pembuat games slot gacor habanero
yang sudah dikenal dari Asia condong Eropa. Habanero berdiri
asal tahun 2012 yang telah mempunyai kantor tetap di Sofia, Manila, Johannesburg, dan Kiev.
Sudah tentu mereka mampu bekerja di jurusan taruhan online teratas tahun 2023.
Ini jadi perhitungan untuk Anda agar langsung mainkan slot gacor
ini hari dari habanero.
Slot Gacor Maxwin PlaynGo
Permainan PlaynGo memberi tubrukan besar di perduniaan slot gacor usai ada pada tahun 2015, apabila pelopor permainan slot yang nyaman dimainkan lewat mobile
Anda, orisinilitas dari PlaynGo memberi kaum games lazim paling penting, yang menghidangkan semua
feature Mengejutkan dan animasi yang elok yang membuat kita ingin terus bermain.
Slot Gacor Maxwin Mimigaming
Games slot gacor Mimi gaming mempunyai tingkat animasi
cepat yang realisitis membuat takjub mata beberapa pemain yang mainkan. Mimigaming mempunyai misi untuk berbobot semua anjuran pengguna
yang harapkan pengertian menyangkut tren games slot gacor terkini yang akan diperlihatkan.
Seriously plenty of fantastic data!
Thanks for finally talking about > What is diabetic ketoacidosis?
– Diabetes Asia Health Magazine < Liked it!
You can certainly see your enthusiasm in the article you
write. The sector hopes for even more passionate writers like you
who are not afraid to say how they believe. At all times follow your heart.